
South Africa might not be the first country that comes to mind as a leading innovator in medical devices, but a number of healthcare companies in the country are changing that perception with a range of innovative medical devices.
According to the Sable Network, a mentoring and consulting services platform for South African companies, South Africa “is a world leader in everything from cell culturing to microsatellites and the technologies of flight.”
On the 3 December 1967, in Groote Schuur Hospital, one of the largest public hospitals in Cape Town, surgeon Christiaan Barnard performed the world’s first human heart transplant and became an overnight celebrity.
Now, South Africa’s healthcare system is digital-ready, the country has medical manufacturing capabilities and knowledge, and a highly competitive, innovative scene distinguishing it from other African economies.
Therefore, developing countries can learn from the way it has sought to modernise its industries.
Here, we profile a range of top medical companies based in South Africa, using 3D-printing and high precision engineering with exotic materials, quickly adapting within the digital transformation era to develop amazing healthcare technologies and services.
Medical devices companies in South Africa
CapeRay
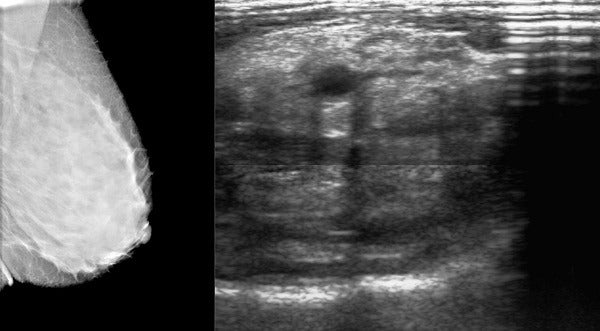
Located in Cape Town, CapeRay is an innovative medical device company launched in 2010 developing several products to help make the process of detecting breast cancer easier for clinicians.
Its first product was named after the Greek goddess of healing, Aceso, a dual-modality system combining full-field digital mammography (a low energy x-ray) and automated breast ultrasound into a single device.
This enables simultaneous acquisition of two sets of breast imaging from two views, improving diagnostic accuracy in dense breasts in just ten minutes.
After it was successfully tested in a clinical trial at the University of Cape Town, the system was launched at Groote Schuur Hospital.
Centre for Rapid Prototyping and Manufacturing

Established in 1997, the Centre for Rapid Prototyping and Manufacturing (CRPM) is located at the Central University of Technology in Bloemfontein and specialises in 3D-printing.
The company is heavily involved with the medical industry and considered to be “one of the top centres in the world and the only one of its kind in the southern hemisphere,” operating ten machines to manufacture prototypes in a number of materials.
This all began in 2007, when titanium laser sintering was introduced in South Africa and CRPM acquired the first machine – one of only six medical imaging machines in the world.
The CRPM currently offers two 3D-printing materials for biomedical applications, to produce pre-operative planning models of a specific patient, a biocompatible plastic material.
The same material can be used to assist surgeons in producing precise surgical aids for cutting or drilling purposes.
Secondly, the CRPM is one of a handful of institutions worldwide that can 3D-print titanium, a biocompatible metal material, to produce patient-specific implants.
DISA Vascular

Situated in Cape Town, DISA Vascular has been developing stents and vascular technology for the treatment of coronary artery disease for the international medical market.
All its products carry the CE mark and have also gained regulatory approval in various other regions outside of Europe.
In 1999, South African biomedical engineer Greg Starke and co-founder Damian Conway launched DISA Vascular – an acronym for “Developed In South Africa,” and set about applying their computational modeling and knowledge of implants to design and build a better coronary stent.
The South African-based research and development, manufactures its products in Germany using up-to-date technology available.
It became the first South African company to gain CE approval of a Class III medical device, Disa Vascular makes about 7,000 devices a year for the treatment of coronary artery disease.
They sell for between R3 000 (£158) and R6 000 (£317).
TiTaMED
Focused on spinal systems, the medical devices company collaborates with surgeons to create new, or improve existing, technologies and techniques to grow the medical industry.
Ti-TaMED has designed its own patented spinal implants that allows for general spinal fusions and a unique scoliosis correction technique, which has CE and US Food and Drug Administration accreditation.
Established in Cape Town in 1996, the manufacturing company specialises in high precision engineering with materials such as titanium, stainless steel, nitronic, aluminium and high performance polymers.
Sinapi biomedical

Founded in 2001, Sinapi biomedical has been developing and manufacturing disposable medical devices such as chest drainage, urinary, obstetrics and feeding devices since 2003.
Sinapi strives to provide devices of excellent quality and reliability, managed by strict and comprehensive quality standards, and plans in the long run to build a production unit in Togo.
Fields of expertise include: Chest drainage, nutrition, urinary drainage and obstetrics.
Sinapi developed a compact chest drainage device that enables greater patient mobility while still maintaining all applications of draining chest fluids after heart, lung and trauma surgery.
The company claims its mechanical (Scheffler) valve is more efficient than existing systems, enabling faster evacuation of blood and air from the chest – shortening chest drainage time – allowing faster removal of the chest catheter and resulting in an improved patient outcome.






