The rapid tests offered by TREKSTOR under the blnk brand are developed and manufactured by established partners from China and are CE-conform according to the EU IVD regulation 98/79/EC
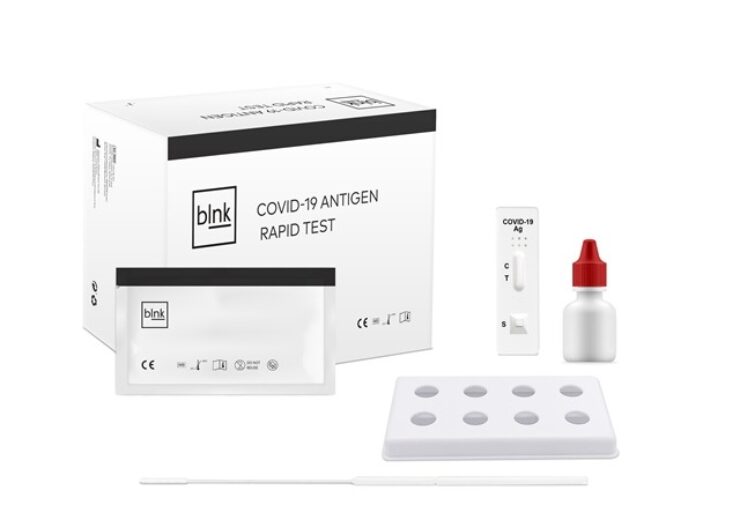
blnk Covid-19 antigen rapid test kit with 20 tests. (Credit: Business Wire)
Given the revision of numerous national testing strategies for countries within the EU, TREKSTOR is happy to announce the availability of its new SARS-CoV-2 Antigen rapid test kit via its healthcare brand blnk.
„Rapid tests will play a vital role to allow extended access to sensible venues like hospitals, care facilities, etc.“, says Shimon Szmigiel, CEO of TREKSTOR. „For the near future we also envision a market in the event space, allowing on-site testing at the venue and therefore providing an extra layer of security.“
The rapid tests offered by TREKSTOR under the blnk brand are developed and manufactured by established partners from China and are CE-conform according to the EU IVD regulation 98/79/EC. The test can be done by having medical professionals take a nasopharynx swab, diluting it in the provided buffer and dripping the resulting solution onto a prepared test-cassette. The result will then be visible after 15 minutes.
The test sensitivity currently is 90,4%, the specificity 99,5%, resulting in 97,6% accuracy. Further clinical studies will improve these values even more over time. Apart from providing a COVID-19 test kit with 20 Antigen tests, TREKSTOR also offeres a combined COVID-19 and Influenza A+B rapid test with 97,5% and 98,8% accuracy.
“Especially during the winter months, symptoms will be misinterpreted“, Shimon Szmigiel explains. „But we also want to make sure that people at risk are not only protected from one danger, but then deliberately exposed to another.“
Source: Company Press Release
