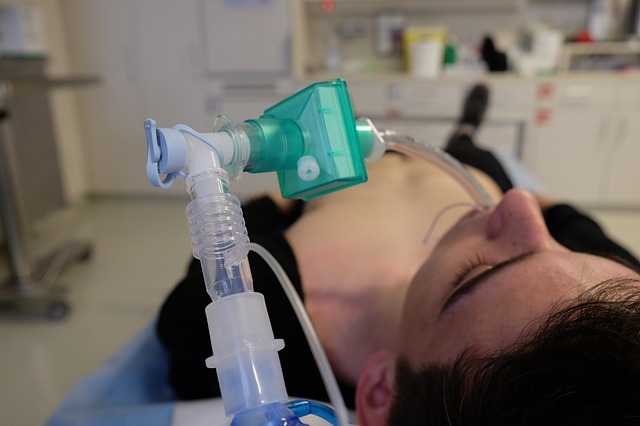
The Health Bank (THB) Global has collaborated with Nanotronics Health for the supply of nHale non-invasive ventilator in the Middle East, North Africa and South Asia (MENASA) region.
Nanotronics Health had secured emergency use authorisation (EUA) from the US Food & Drug Administration for nHale ventilator to be used in traditional healthcare facilities and areas transformed for the care of large numbers of Covid-19 patients.
The non-invasive ventilator can also be used in at-home settings with a doctor’s prescription.
The authorisation also enables the optional use of supplemental oxygen in combination with nHale ventilator based on the suggestion of a medical professional.
THB Global strategy head Zarmina Jafar said: “In pursuit of finding a solution for managing COVID-19 symptoms through non-invasive methods, we have partnered with Nanotronics Health, LLC, for the sale and distribution of the nHale.”
Non-invasive ventilation is essential for Covid-19 patients in the hospital. To meet this requirement, Nanotronics Health had leveraged its parent company Nanotronics’ in-house expertise to integrate advanced artificial intelligence (AI), intelligent factory control (IFC) and modern engineering capabilities for the designing and manufacturing of nHale ventilator within 90 days.
The nHale ventilator has been designed to use in non-life-threatening situations
Designed to be used in non-life-threatening situations, the nHale bi-level positive air pressure device helps deliver respiratory therapy for spontaneously breathing adults weighing more than 30kg and suffering from Covid-19 disease.
The Health Bank will also provide training to medical professionals for efficient delivery of non-invasive respiratory therapy to patients in the region.
Nanotronics Health president Julie Orlando said: “We developed nHale to meet the urgent need for non-invasive respiratory options for patients suffering from Covid-19.
“We designed for quality, comfort and ease of use, making nHale available at a fraction of the cost of other non-invasive ventilators.”
In June, Enexor Health Systems secured FDA EUA status for its new X-VENT ventilator, which will help deliver the critical modes of ventilation needed for Covid-19 patients.






