Asahi Kasei Group company Zoll Medical has acquired US-based medical device firm TherOx for an undisclosed sum.
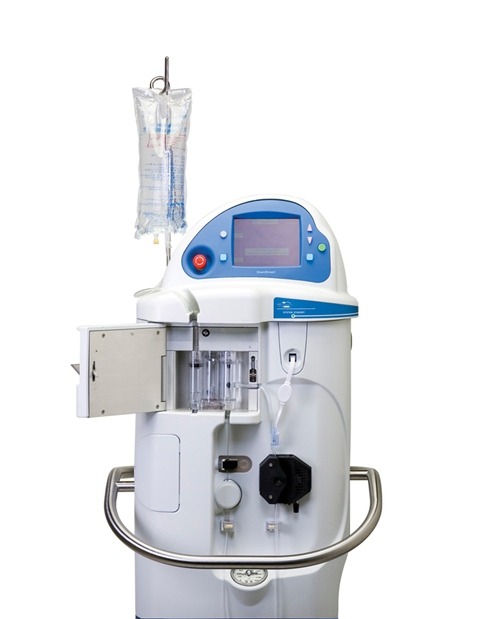
Image: SuperSaturated Oxygen Therapy. Photo: courtesy of Business Wire.
Based in Irvine of California, TherOx is involved in the development and manufacturing of dissolved oxygen delivery mechanisms and systems to improve the standard of care in heart attack treatment.
The company is primarily focused on improving treatment of acute myocardial infarction (AMI) and markets systems to deliver SuperSaturated Oxygen (SSO2) Therapy.
Zoll Circulation president Neil Johnston said: “This acquisition expands the ZOLL product portfolio in support of excellence for patients and caregivers.”
SSO2 Therapy, which compliments primary coronary intervention (PCI) for acute heart attack, has been developed to restore microvascular blood flow and reduce damage to the heart muscle, helping to improve overall heart function.
Recently approved by the Federal Drug Administration (FDA), the SSO2 Therapy has been shown to reduce infarct size after PCI with stenting in patients with left anterior descending ST-elevation myocardial infarction.
SSO2 Therapy is produced using in-hospital oxygen and saline for the creation of a superoxygenated solution. It will be mixed with the patient’s arterial blood to create superoxygenated blood.
SSO2 Therapy delivers a one-time and 60-minute infusion of the patient’s super oxygenated blood to the targeted ischemic area of the heart through a small catheter, immediately after the coronary artery has been opened by PCI.
The therapy infuses super oxygenated blood to enhance microvascular flow and then restores heart tissue to normal oxygen level.
TherOx president and CEO Kevin Larkin said: “SSO2 Therapy can help reduce infarct size among patients who suffer from large anterior AMIs, which are the most serious heart attacks.
“We are looking forward to becoming part of ZOLL so that together we can bring this breakthrough treatment option to more patients sooner.”
Zoll Medical is engaged in the development and marketing of medical devices and software solutions for defibrillation and monitoring, circulation and CPR feedback, data management, therapeutic temperature management, and ventilation.
