Both cardiac ultrasound solutions have secured CE mark approval, as well as received 510(k) clearance from the US Food and Drug Administration (FDA).
Philips’ EPIQ CVx cardiovascular ultrasound system includes higher processing power and advanced image clarity and sharpness, as well as improved exam efficiencies and reproducible quantification enabled by anatomical intelligence.
The system will also feature TrueVue technology that allows clinicians to view photorealistic renderings of the heart,
TrueVue technology enables to improve cardiac anatomy analysis by providing detailed tissue and depth perception imaging through a new virtual light source.
Based on dynamic heart model, the system uses anatomical intelligence to automatically quantify left ventricle function to produce a multi-beat analysis for adult patients.
In addition, the system features cardiology-specific user interface, which simplifies the exam experience through a user-configurable touch-screen interface. It helps clinicians to personalize their controls and improve workflow for their cardiology exams.
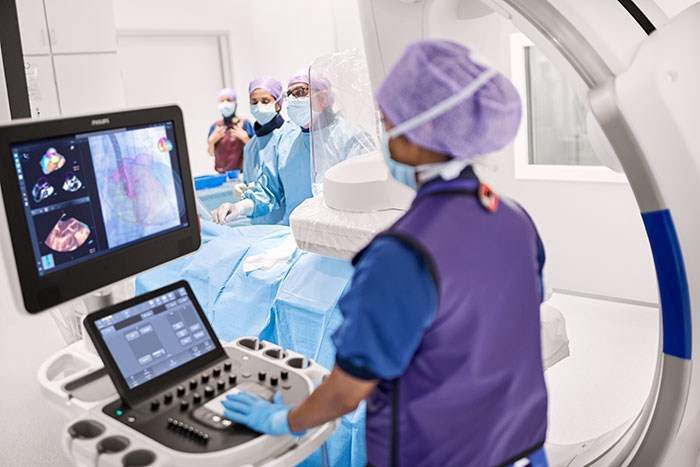
Developed for use in the cath lab, the the EPIQ CVxi with EchoNavigator is the firm’s third-generation interventional solutionto, which will streamline communication between the interventional cardiologist and the echocardiographer during complex interventional exams.
Through combining live ultrasound and X-ray information into one view, EchoNavigator enables interventional cardiologists to monitor procedures along with the location of crucial anatomical structures.
Philips will exhibit the both cardiac ultrasound solutions at this year’s European Society of Cardiology event in Munich, Germany.
Philips cardiac ultrasound general manager David Handler said: “With the EPIQ CVx and its cardiology-centric design, clinicians can perform fast exams with enhanced diagnostic information.
“The EPIQ CVxi is our third-generation integrated ultrasound-angiography cath lab solution for real-time, workflow-optimized image guidance and advanced quantification for structural heart procedures.”
Based in Netherlands, Royal Philips is engaged in the development of diagnostic imaging, image-guided therapy, patient monitoring and health informatics solutions.
With around 75,000 employees, the company carries out operations in more than 100 countries.






