Mevion Medical Systems has been the leading provider of compact proton therapy systems for cancer care
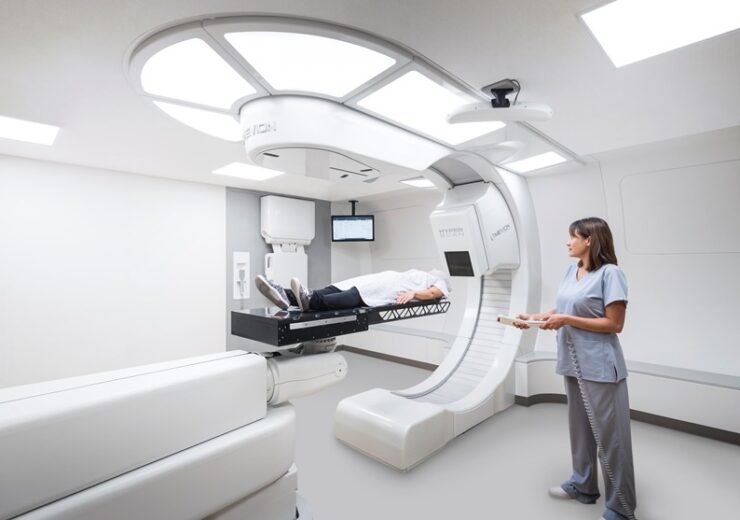
The MEVION S250i Proton Therapy System with HYPERSCAN Pencil Beam Scanning. (Credit: Business Wire.)
Mevion Medical Systems and Proton International announced today that they have signed a two-system contract to bring Mevion’s compact proton therapy solution to new centers in locations to be announced in South Texas and the Southeast United States.
The new centers will feature the MEVION S250i Proton Therapy System® with HYPERSCAN® Pencil Beam Scanning (PBS). HYPERSCAN enables faster and sharper delivery of therapeutic radiation to tumors. The system’s leading-edge clinical capabilities, combined with its compact, affordable design, and industry-leading ramp-up time, has changed the landscape of proton therapy. Today, more cancer centers are considering providing compact proton therapy to their patients because of the technology Mevion has advanced.
“We selected Mevion’s compact system because it has the ability to offer an efficient clinical solution adopted by many major cancer programs while minimizing costs,” said Chris Chandler, chief executive officer of Proton International. “PI specializes in providing custom single-room proton therapy solutions to our clinical providers, and this technology will be a powerful addition to their cancer treatment arsenal.”
“Proton International’s successful approach to proton therapy by focusing on building and operating centers that support their customer’s goals made them a natural partner,” said Tina Yu, Ph.D., chief executive officer of Mevion. “We look forward to collaborating closely with Proton International on these new centers and providing greater access to this lifesaving technology to patients in Texas and the Southeast.”
As the leading supplier of compact proton therapy systems in the United States, Mevion has been selected by more NCI-Designated Cancer Centers than any other manufacturer. In July, another MEVION S250i Proton Therapy System began treating patients at Barnes-Jewish Hospital in St. Louis, MO, and Huntsman Cancer Institute at the University of Utah in Salt Lake City, Utah will be clinically operational at the end of 2020.
Source: Company Press Release
