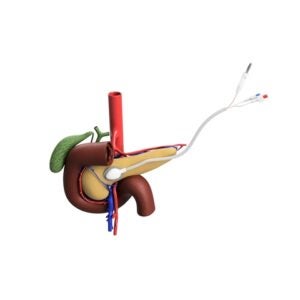
Medtronic’s Gastrointestinal and Hepatology business, which is part of the Minimally Invasive Therapies Group and Q3 Medical agreed to a six-year, exclusive agreement to distribute ARCHIMEDES biodegradable stent in western EU, US, and Japan; with rights to other regions. Q3 Medical will continue to expand its presence in South East Asia, Latin America, Eastern Europe, and the Middle East.
Typically, biliary and pancreatic duct stents are made of plastic or metal, however the ARCHIMEDES stent is made of a combination of dissolving materials permitting different rates of degradation depending on the patient need. Use of degrading materials in a stent, decreases reinterventions that are common with current stents and allows the potential for eliminating a stent removal procedure which is almost always done for plastic and metal stents currently. ARCHIMEDES has CE Mark.
Eric Mangiardi, President & CEO of Q3 Medical stated, “We are excited and fortunate to have partnered with a world leading medical device organization like Medtronic. Entering into this agreement is a major step toward our goal of becoming the world leading developer of biodegradable implants. Mangiardi further stated; “The promise of ARCHIMEDES is based in its potential to eliminate the second removal procedure required for traditional plastic stents, which could reduce billions of expenses from the global healthcare system while improving patient care.”
Source: Company Press Release