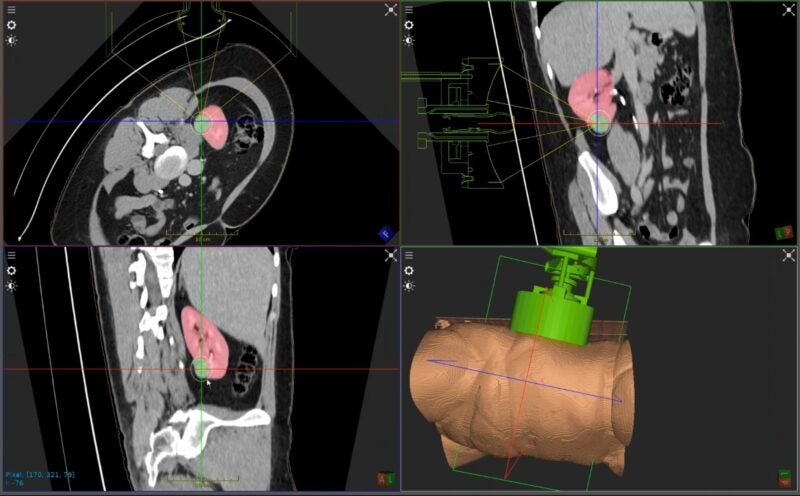
US-based medical device company HistoSonics has announced treatment of the first kidney patient using its histotripsy non-invasive platform and sonic beam therapy.
The first treatment, which was for a patient with primary solid renal tumour, was conducted in Leeds, UK, by Tze Min Wah, senior consultant radiologist and clinical lead for interventional oncology program at the Leeds Teaching Hospitals NHS Trust.
According to HistoSonics, the latest procedure represents the initial treatment in the HistoSonics sponsored CAIN trial.
The trial is a phase 1 prospective, multi-centre study with a goal to assess the safety and technical viability of the histotripsy system in non-invasive targeting and removing of primary solid renal tumours without the use of incisions or needles.
HistoSonics president and CEO Mike Blue said: “This first treatment is a significant milestone for the Company as it represents expansion into our second active clinical application (after liver) and supports our mission to deliver histotripsy to patients who may potentially benefit from its unique capabilities.
“Our goal is to enable physicians to precisely target and destroy kidney tumours with our novel, noninvasive solution, avoiding the morbidity and complications seen with current invasive surgery or ablative techniques.”
Unlike current kidney therapies, non-invasive histotripsy platform may avoid bleeding and infection, said HistoSonics.
Besides, the company’s platform can potentially destroy targeted tissue without harming non-targeted kidney tissue.
Apart from that, histotripsy’s mechanical mechanism of cellular destruction can retain function of the kidney’s urine collecting system and remove some complications associated with the prevailing invasive approaches.
HistoSonics’ platform is said to use advanced imaging and sensing technology to provide tailored treatments with precision and control.
During the procedure, the targeted tissue is mechanically destroyed and liquefied at subcellular levels using concentrated sound energy to create controlled acoustic cavitation, the medical device firm added.






