eSuction is the only distal cap device on label for removal of food bolus impactions and provides an efficient solution for the removal of impactions, which physicians have found commonly accompany some type of pathology
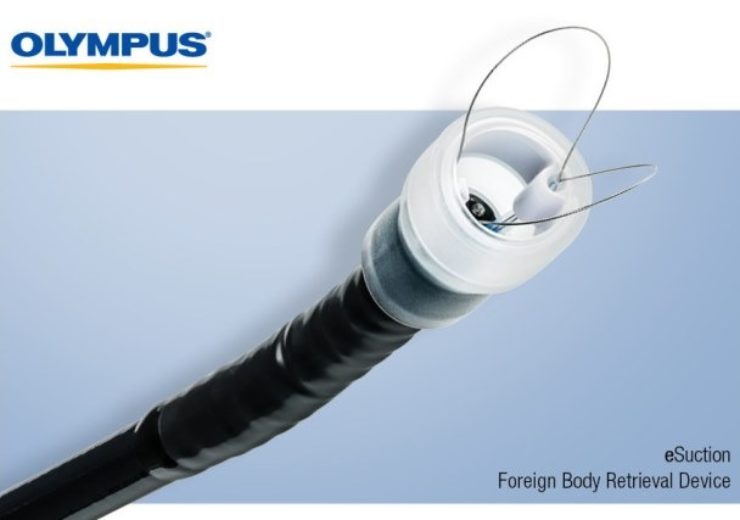
Image: Olympus is the exclusive U.S. distributor of eSuction from EndoTherapeutics. Photo: courtesy of Olympus / PRNewswire.
Olympus, a global technology leader in designing and delivering innovative solutions for medical and surgical procedures, among other core businesses, announced today the U.S. launch of eSuction for removal of foreign bodies.
eSuction from EndoTherapeutics combines snare technology with suction to aid in the retrieval of food bolus impactions, foreign bodies and excised tissue such as polyps. Through an exclusive distribution agreement with Olympus, eSuction is now available in the U.S.
eSuction, which is FDA registered as a Class I medical device, is designed to fit securely onto the endoscope.
eSuction is the only distal cap device on label for removal of food bolus impactions and provides an efficient solution for the removal of impactions, which physicians have found commonly accompany some type of pathology.[i] It brings economic value in that it may reduce the need for an overtube in the removal of sharp foreign bodies, and it may aid in reducing procedural time by providing a combination device that limits the need for device exchange. With the addition of eSuction, Olympus continues to offer one of the most diversified portfolios for foreign body retrieval.
“Our EndoTherapy line is one of the most exciting of the Olympus businesses today,” said Kurt Heine, Group Vice President at Olympus America Inc. “We are pleased to be working with EndoTherapeutics to distribute the eSuction device, an important addition to the Foreign Body Removal line in terms of cost-savings and clinical benefits. This is an important device for our customers and their patients.”
eSuction is being made available to customers immediately and will be on display in Olympus Booth #502 at NASPGHAN, October 17-19, at the Sheraton Grand in Chicago.
Source: Company Press Release
