The company's ActivShield is a patent pending advanced technology capable of effectively disinfecting FFP2 masks
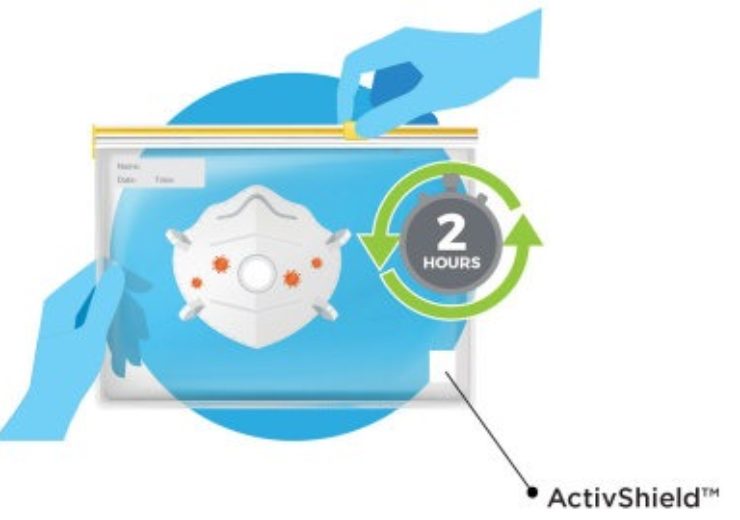
FFP2 mask decontamination with ActivShield by Aptar (Credit: Aptar.)
US-based drug delivery solutions provider AptarGroup is seeking an Emergency Use Authorisation (EUA) from the German Federal Institute for Drugs and Medical Devices (BfArM) for its ActivShield solution to disinfect FFP2 filtering facepiece respirators (FFP2 masks).
ActivShield is a patent pending advanced technology capable of effectively disinfecting FFP2 masks used by healthcare workers and first responders.
The FFP2 masks are very much needed by healthcare professionals, amid the worldwide shortage of disposable masks during the Covid-19 pandemic, said the company.
Aptar president and CEO Stephan Tanda said: “We are proud to continue to support the fight against the COVID-19 pandemic by furthering our efforts to bring this innovative technology to the global health care community.
“We have a long-standing presence in Germany and we will continue to support the communities where we live and work.
“Our technology provides a unique, simple, and effective way to help solve the critical problem of PPE shortages we’re currently facing. We look forward to working with the U.S. FDA and BfArM to bring ActivShield™ to market and fulfill the ongoing unmet need.”
Aptar uses chlorine dioxide as disinfectant in its ActivShield technology
Aptar said that its ActivShield technology is a simple disinfecting process, where the FFP2 mask and a small strip of decontaminant are placed inside a commonly available one-gallon plastic bag.
The strip releases controlled amount of chlorine dioxide to decontaminate the FFP2 mask inside the sealed bag, and the process would take only two hours until the mask is ready to wear again.
Chlorine dioxide is widely used as a disinfectant in different industries, including the paper industry, drinking water treatment, food processing, and medical equipment.
The delivery mechanism of the ActivShield technology leverages the disinfecting properties of chlorine dioxide in a controlled sustained release within a contained volume. It can be performed on-site at the local hospital where the mask is being used.
Aptar said that the EUA submission of its technology in Germany is part of its plans to expand the global access to FFP2 respirators, facilitating decontamination and reuse of masks.
In addition, the EUA submission with BfArM follows the submission for US FDA Emergency Use Authorisation in April 2020.
The company is engaged in designing and manufacturing a range of drug delivery, consumer product dispensing and active packaging solutions.
