Proclaim XR Apple mobile digital devices and Bluetooth wireless technology to manage pain and fit seamlessly into patient’s lifestyle
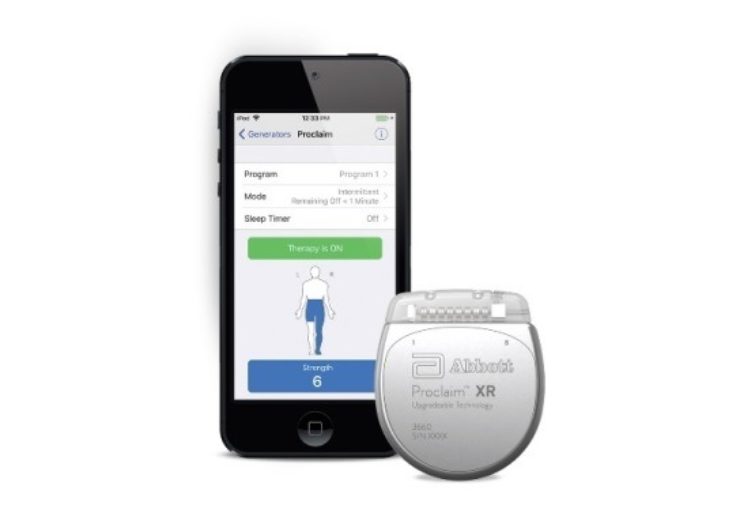
Image: Abbott’s Proclaim XR with programmer. Photo: courtesy of Abbott.
Abbott has secured approval from the US Food and Drug Administration (FDA) for its Proclaim XR recharge-free neurostimulation system to treat people with chronic pain.
Proclaim XR platform provides a low dose of Abbott’s advanced stimulation waveform, which was developed based on scientific insights from doctors and research to mimic natural patterns observed in the brain.
The low doses of mild electrical pulses will be used by the platform to change pain signals, as they travel from the spinal cord to the brain.
The delivery of lower doses of spinal cord stimulation (SCS) will help expand the battery life of the system.
Proclaim XR system is under assessment for CE mark approval
Abbott developed the Proclaim XR system based on positive results from its BurstDR micrOdosing stimuLation in De-novo patients (BOLD) study.
The trial demonstrated 100% of 24 recruited patients on a low-energy BurstDR dosing programme experienced pain relief with less than six hours of battery use per day, while around 50% of those patients achieved pain relief with the lowest effective dose.
The Proclaim XR system enables physicians to detect the lowest effective dose of BurstDR stimulation customised to each patient.
At present, the Proclaim XR system is under assessment for CE mark approval.
Abbott’s neuromodulation business vice president Keith Boettiger said: “Proclaim XR is the latest advancement in patient-centric pain therapy — opening the doors for people who don’t want to be burdened with the hassles of recharging. Recharge-free means patients can live their lives without the constant reminder of being in treatment.
“This is a meaningful advancement for the millions of people who need new options when it comes to managing the effects of their chronic pain. Abbott is committed to developing life-changing solutions and putting the patient at the center of everything we do.”
Recently, Sanofi and Abbott have announced a partnership to integrate glucose sensing and insulin delivery technologies to help people with diabetes manage their condition.
