Netherlands-based Microsure has closed its Series B2 investment round to secure €38m in development capital to advance and finalise the development of MUSA-3, its latest microsurgical robot.
The funding round included new investors that included the European Innovation Council Fund (EICF), kineo, Invest-NL, and other private investor groups.
It was supported by key existing shareholders including BOM Ventures, Innovation Industries, private investors Ten Cate Investerings Maatschappij (TCIM), and others.
The Dutch company also signed an asset financing agreement with kineo to lease MUSA-3 to hospitals.
Microsure intends to use the funds to progress its robot to the clinical trials phase, followed by regulatory clearance in the US and Europe.
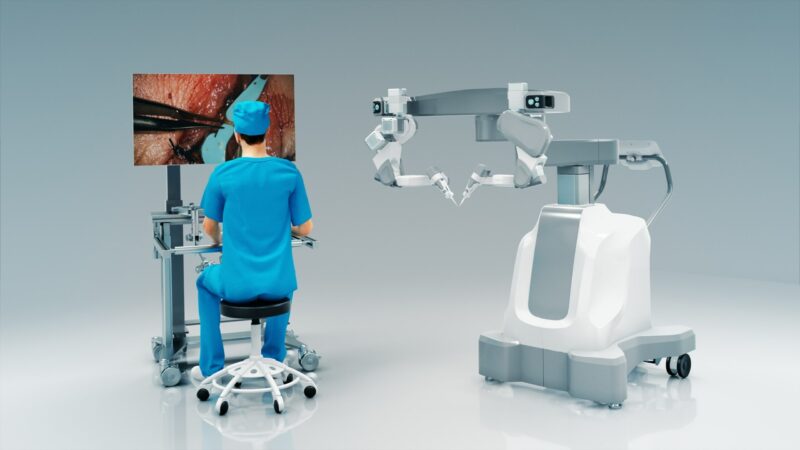
MUSA-3, the firm’s first commercial product, is designed for open microsurgery. It is said to empower surgeons with enhanced precision, accuracy, stability, and control to enable new interventions that are currently difficult to do by hand.
The system builds upon its previous generations, the MUSA-2 robot, to provide enhanced dexterity and a wide workspace and tremor reduction.
According to Microsure, MUSA-3 can be integrated easily with surgeons’ current micro-instruments of different brands.
Adaptable to a variety of surgical operations, the microsurgical robot combines surgeon comfort with improved ergonomics to enable physicians to work from a console with digital exoscopes or hybrid surgical microscopes.
Microsure CEO Sjaak Deckers said: “The tireless efforts of the Microsure team over the past years have culminated in the development of a truly unique robot celebrated for its precision, ergonomic design, and adaptability for a wide spectrum of intricate procedures.”
“Our progress is a testament to the exceptional talent within our growing team of dedicated individuals and the seamless collaboration with our key development and manufacturing partners.
“The continued expansion of Microsure’s clinical network strategically positions the company for upcoming clinical studies, an integral part of our journey towards securing FDA clearance and CE-mark certification.”
The development of MUSA-3 was based on the positive results and valuable insights from multiple clinical studies of MUSA-2.
The trials were conducted at the Maastricht University Medical Center in the Netherlands and Uppsala Academic Hospital in Sweden.






