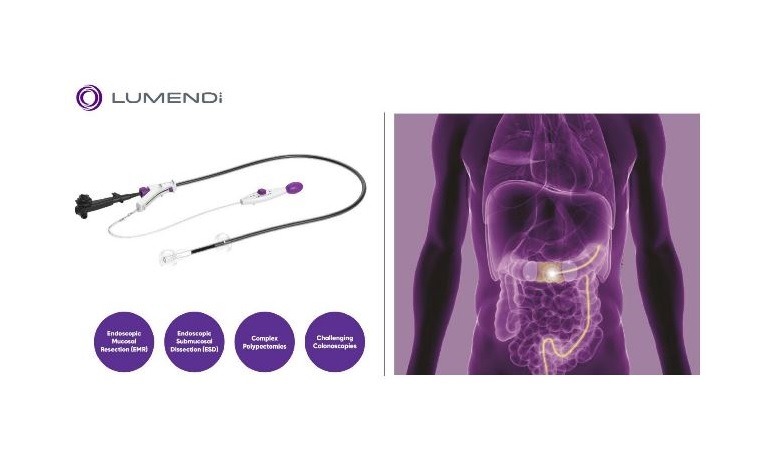
US-based medical devices maker Lumendi has introduced a new hydrophilic coating called EZ Glide for its DiLumen endoluminal interventional platform (EIP).
The new hydrophilic coating has been developed to apply for the inner sheath of DiLumen EIP, a double-balloon endoluminal device.
DiLumen EIP will help safely enhance the navigation of the endoscope via the bowel to create a stabilised therapeutic zone inside the colon during endoluminal interventions and reduce potential mucosal injury.
The activation of EZ Glide is done by water or saline to improve the endoscopic performance of the DiLumen EIP by further stabilising the endoscope tip and nearby anatomy.
It will also provide gentler anatomic control to safely straighten and navigate tortuous anatomy for enhanced control and quicker access to lesions.
EZ Glide will also help avoid the use of messy gels or oils during preparation and further enhances intraoperative visibility.
In addition, it is designed to facilitate safe and repeated access as a conduit to efficiently exchange tools and tissue removal.
Lumendi CEO Dr Peter Johann said: “The DiLumen EIP has the potential to improve or enable less invasive procedures for benign colorectal polyps, and can take the place of open or laparoscopic surgery, thereby preserving anatomy, shortening recovery and reducing healthcare costs.”
In August 2019, Lumendi secured 10(k) clearance from the US Food and Drug Administration (FDA) for its DiLumen lk endolumenal interventional knife.
DiLumen lk endolumenal interventional knife is a sterile, single-use and disposable monopolar electrosurgical device designed to be used for cutting, dissecting and cauterising tissue within the digestive tract during endoscopic procedures.




