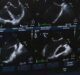
Lantheus secures FDA approval of VIALMIX RFID to activate DEFINITY
Lantheus Holdings has secured the US Food and Drug Administration (FDA) supplemental new drug application (sNDA) approval for VIALMIXRFID device, for use with DEFINITY Vial. The medical equipment