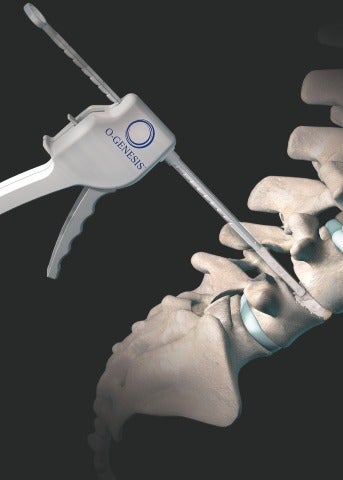
Orthofix Medical, a medical device firm focused on spine, has rolled out its new O-GENESIS graft delivery system to deliver allograft, autograft or synthetic bone graft to orthopedic surgical sites.
Along with the O-GENESIS System, the company is also launching the AlloQuent Structural Allograft Q-Pack, a hydrated cervical and lumbar spacers for allograft procedures.
The medical device maker said that its O-GENESIS graft delivery system works closely with its advanced biologic solution, Trinity ELITE Allograft with viable cells, processed for MTF Biologics.
With the ergonomic design, the system is said to enable unassisted delivery, with two easy loading options to facilitate a flexible choice of other bone graft materials, including fiberFUSE Demineralized Bone Matrix.
Orthofix partnered with MTF Biologics to launch AlloQuent Structural Allograft
AlloQuent Structural Allograft is a precision-machined cervical and lumbar spacers and Orthofix has partnered with MTF Biologics to launch AlloQuent in the new Q-Pack Technology.
Q-Pack is a zero rehydration technology designed to reduce the preparation time and preserve the mechanical properties of the allograft, said the company.
Orthofix is a Lewisville, Texas-based medical device and biologics company with a spine and extremities focus. Its products are distributed in more than 70 countries.
Orthofix Spine global president Kevin Kenny said: “Orthofix is committed to continuous technology innovation and expanding solutions to improve operating room efficiencies.
“The unique design of the O-GENESIS Graft Delivery System enables one-handed delivery of bone graft material in a precise and controlled manner to aid surgeons with the targeted placement of biologics during orthopedic surgeries.
“Additionally, we are pleased to offer the new AlloQuent Structural Allograft in Q-Pack that allows for immediate use with no downtime for rehydrating. Both of these demonstrate our commitment to providing surgeons with easy-to-use solutions to meet the needs of their patients.”
The JuniOrtho plating system, which is available in a range of plate sizes with different lengths, accommodates both locking and non-locking screws corresponding to the plate size.






