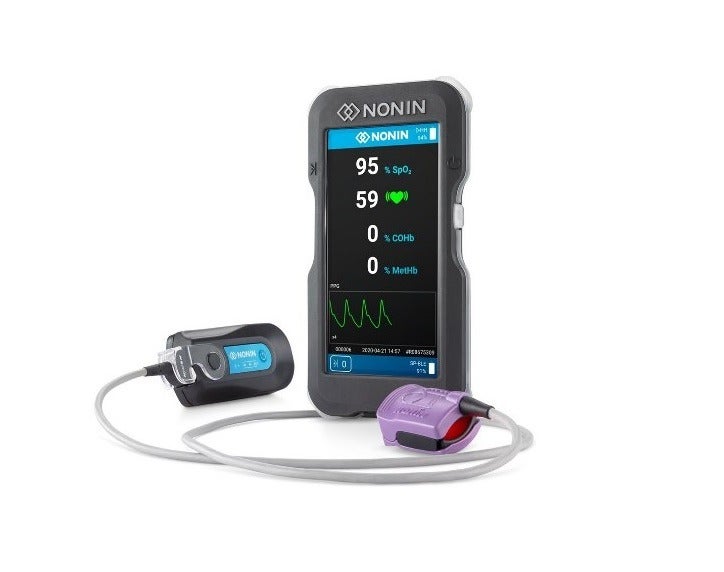
Nonin Medical has secured 510(k) clearance from the US Food and Drug Administration (FDA) for its CO-Pilot wireless handheld multiparameter system (H500).
The handheld device with wireless capabilities will enable EMS, firefighters, and military personnel to measure patient parameters following cardiac arrest, traumatic injury, carbon monoxide or smoke inhalation.
Nonin’s CO-Pilot system has the capacity to deliver stable readings to provide better patient care in an ambulance and at the scene of a fire, as well as in any emergency setting before arrival at a hospital.
The handheld device is said to simultaneously track patient pulse oximetry (SpO2), pulse rate, carboxyhemoblin (COHb), and methemoglobin (MetHb). An additional sensor can be used to measure cerebral and tissue oximetry (rSO2).
CO-Pilot system can measure brain and tissue oxygenation when administering CPR to patients who experience out-of-hospital cardiac arrest (OHCA), said the company.
Nonin’s system delivers tissue oximetry data for the tracking of decreases in tissue oxygenation, which is a critical metric for survival and brain function.
The handheld device facilitates the clinical evaluation of carbon monoxide poisoning
The handheld device is also helpful in the clinical evaluation of carbon monoxide poisoning, enabling first responders to make treatment decisions at the scene or on the way to a hospital.
According to the company, the device allows first responders to efficiently measure SpO2, pulse rate, COHb, MetHb, and rSO2 even in the most challenging conditions.
Nonin Medical CEO Dave Hemink said: “Since 1986 when Nonin advanced pulse oximeter technology, the company has set the standard for innovation and accuracy.
“The CO-Pilot is another Nonin industry first that leverages wireless technology to give first responders potentially life-saving patient data to decide on treatment options during the golden hour.”
In 2016, Nonin Medical introduced the next generation of end-tidal CO2 monitors in the LifeSense II capnograph/pulse oximeter and RespSense II capnograph.
Nonin Medical offers durable noninvasive patient monitoring devices for healthcare professionals and consumers. Its product portfolio is comprised of pulse oximeters, cerebral and tissue oximeters, capnographs, sensors, and software solutions.






