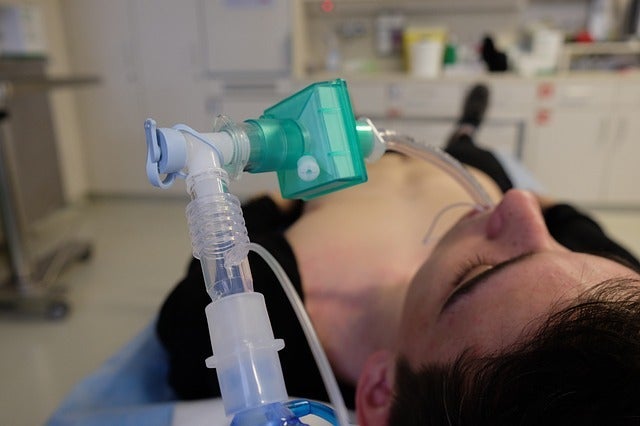
Nanotronics Health has selected Solutions in Critical Care (SCC) to distribute its nHale non-invasive respiratory support device, which is designed for the treatment of Covid-19.
The non-invasive respiratory support device has secured emergency use authorisation from the US Food and Drug Administration (FDA) to treat Covid-19 patients in the hospital and spaces converted to treat Covid-19 patients, as well as in private homes within the US.
nHale, a bi-level positive air pressure device, has been designed to support spontaneously breathing adults weighing more than 30kg suffering from Covid-19 disease.
Nanotronics’ machine is suitable for use in traditional healthcare facilities such as hospitals, assisted living facilities and nursing homes, as well as converted spaces such as convention centres, university dormitories and hotels.
According to the company, supplemental oxygen may be used with the nHale device to enhance the oxygen concentration of the airflow being provided to the patient only when prescribed and trained by a qualified medical professional.
The warnings have to be checked when using supplemental oxygen with the nHale device, said the company.
Nanotronics Health president Julie Orlando said: “We are pleased to partner with an established distributor who understands the need to deliver accessible devices to patients and healthcare professionals when they need it.
“As hospitals continue to be strained throughout the country, it is our hope that we can provide a solution to both ease that strain while also providing reliable at-home support.”






