The infusion set has been designed to extend the length of time to help users stay on insulin pump therapy for a long time
Medtronic Extended infusion set. (Credit: Medtronic)
Medical technology firm Medtronic has introduced its infusion set in select European countries to help minimise burden for diabetes people.
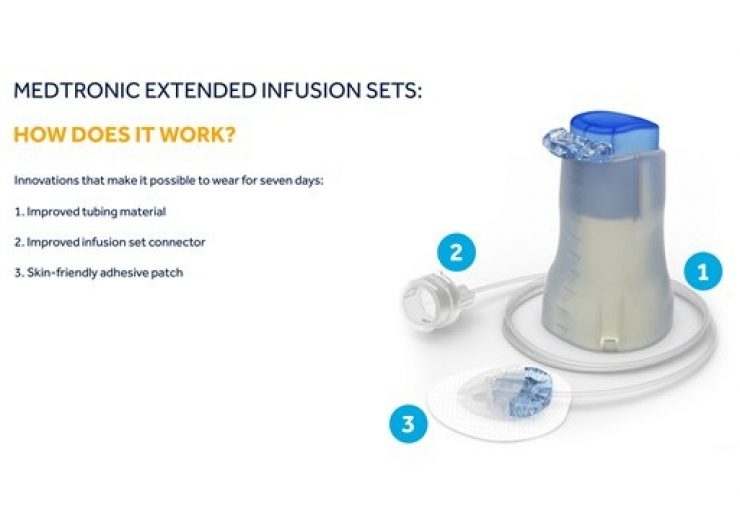
Claimed to be the world’s first and only infusion set, the Medtronic Extended infusion set is designed to be worn for up to seven days.
Medtronic has worked with ConvaTec’s subsidiary Unomedical for the development of the new infusion set.
An infusion set is tubing designed to offer insulin from the pump to the body. The process generally needs a set change every few days.
The Medtronic Extended infusion set has been designed to extend the length of time to help users stay on insulin pump therapy for a long time with fewer interruptions and insertions.
The infusion set is said to be compatible with all MiniMed 600 and 700 series insulin pumps.
The infusion set’s new design leverages advanced materials, which enables to minimise insulin preservative loss and maintain insulin flow and stability.
The new tubing connector enhances the physical and chemical stability of insulin and authenticity of infusion site performance, in addition to reducing the risk of infusion set occlusion.
The adhesive patch features an adhesive layer, which expands wear time and offers comfort and durability for up to seven days.
The Medtronic Extended reservoir is also evaluated and approved to keep insulin stable and safely used for up to seven days, said the company.
Medtronic diabetes business customer experience vice president Julie Foster said: “Our goal with this latest innovation is to make insulin pump therapy easier for people living with diabetes.
“The Medtronic Extended infusion set introduces increased simplicity and convenience — something we’re looking to deliver across all of our products and services to help ease burden and enhance the experience for people wherever they are in their diabetes journey.”
Medtronic is offering the infusion set in select European countries such as Finland and Belgium, as well as plans to launch in other European countries this year via a phased approach.
The infusion set is used for investigation purposes only and not approved for sale or distribution in the US.
