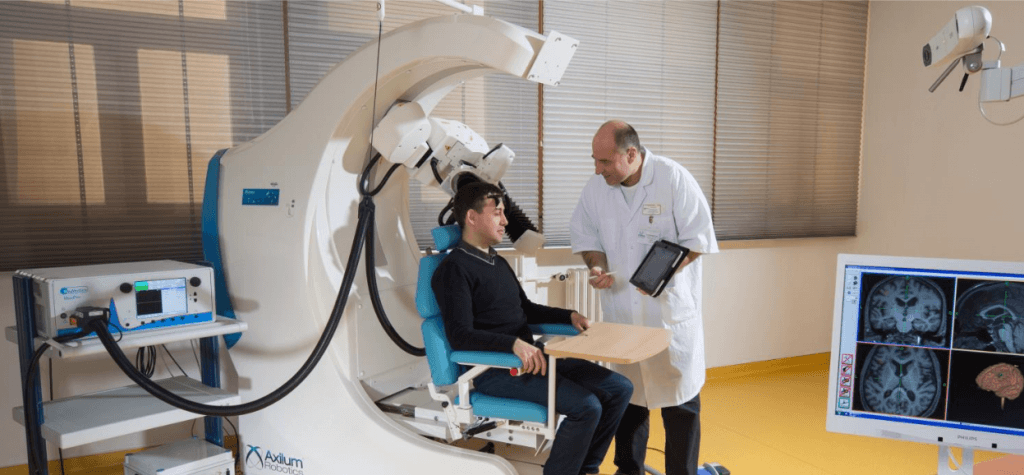
MagVenture said that the TMS-Cobot is a new solution that is expected to help further elevate the rapidly growing field of neuromodulation. It is more commonly known as TMS or transcranial magnetic stimulation.
The medical device company claims that the TMS Therapy is an effective, non-invasive treatment option for the patients not responding to antidepressants.
The treatment using TMS Therapy involves application of magnetic pulses to a particular area of the patient’s brain with the use of a magnetic coil. The particular spot is the entry-point for the treatment of depression.
The magnetic pulses reach the neural brain network controlling mood and emotion, and are expected to alleviate the depression.
MagVenture sales VP Kerry Rome said: “While our customers express a very high level of satisfaction with the quality, cost-effectiveness and longevity of the MagVenture TMS Therapy solution, some are also ready to take their TMS practice to the next level, so to speak, without compromising on quality or safety.
“Adding a robotic arm has two major advantages for these customers: Firstly, it provides a continuous head motion tracking system which actively follows the patient’s possible head movement during treatment. Secondly, it will reduce the manual coil handling time for the TMS operator which especially is a key factor for clinics with a high number of patients.
Furthermore, the patient may fully awake or resume to his or her usual activities right after the TMS treatment session and typically a patient is expected to receive one per weekday, to make 20-30 treatment sessions in total.
Rome added: “The TMS-Cobot is a good example of how the field of TMS is constantly evolving. We, as a TMS provider, need to stay tuned to meet customer and patient demands, for today and tomorrow.”
Axilum Robotics, a France-based medical robotics company, has developed the TMS-Cobot. The device is FDA indicated for the spatial positioning and orientation of the treatment coil of the MagVenture TMS Therapy system.
Axilum Robotics CEO Michel Berg said: “We are very happy to partner with MagVenture who will distribute our device in the U.S. along with its MagVenture TMS Therapy system. We are convinced that this robotized system represents a major step forward for the implementation of TMS in the USA.”
MagVenture claims that it is the first TMS company in 2018, to receive FDA clearance for a much shorter protocol called Express TMS, and reduces the treatment time from the standard 37 minutes to just 3 minutes per session.
The company said that its TMS Therapy is specified only for the treatment of Major Depressive Disorder in adult patients who have failed to receive satisfactory improvement from prior antidepressant medication in the current episode.






