With the approval, Monarch platform becomes the first and only multi-speciality, flexible robotically assisted system designed for use in both bronchoscopy and surgical removal of kidney stones
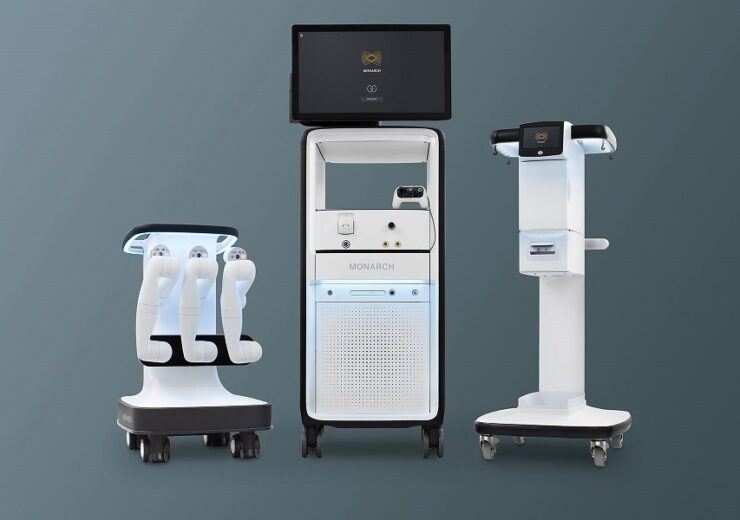
Monarch is a robotic platform for endourological procedures. (Credit: Auris Health, Inc.)
Auris Health, a subsidiary of Johnson & Johnson MedTech company Ethicon, has received the US Food and Drug Administration (FDA) 510(k) approval for its Monarch platform to enable endourological procedures.
With the FDA approval, Monarch platform becomes the first and only multi-speciality, flexible robotic solution for use in both bronchoscopy and urology.
Ethicon said that it has designed the robotic platform to help urologists precisely reach and visualise areas within the kidney with superior control.
The company intends to commence a first-in-human clinical study to evaluate the Monarch platform’s endourologic application later this year.
Ethicon Group chairman Vladimir Makatsaria said: “This latest FDA 510(k) clearance for Monarch delivers on our vision to extend the robotic platform’s capabilities across multiple specialities, enabling hospital systems to target two disease states using one device.
“At Ethicon, we’re committed to driving meaningful innovation that elevates the standard of care for patients and delivers improved solutions that address important clinical challenges for our customers.”
According to the company, Monarch has been designed to provide urologists with a single platform to support both ureteroscopic and percutaneous nephrolithotomy (PCNL) procedures.
Ureteroscopic procedures are the most commonly performed surgical stone procedures worldwide, but become highly challenging with the increase in stone size.
PCNL procedures have proved to provide superior stone clearance for patients with larger kidney stones. However, they represent only 7% to 8% of stone procedures conducted in the US to date, as various factors hinder the frequent use of PCNL
Ethicon claimed that its Monarch will address most of those complications by deploying unique and minimally invasive technology.
Several physicians have been using the robotic platform since 2018, for assisted bronchoscopy procedures and to reach small, hard-to-reach peripheral lung nodules at an earlier stage, with enhanced precision.
University of Southern California clinical urology professor Mihir Desai said: “Monarch reduces the complexity of gaining high-quality percutaneous access and aids stone clearance efficiency through simultaneous fragmentation and suctioning of stones with robotic assistance.
“With this platform, many urologists may be willing to expand their practice to include percutaneous access and PCNL procedures, thereby increasing patient access to more effective treatments closer to home.”
University of California Irvine department of urology professor Jaime Landman said: “With this FDA clearance, Monarch is poised to aid physicians in their goals of reducing overall retreatment and complication rates.”
