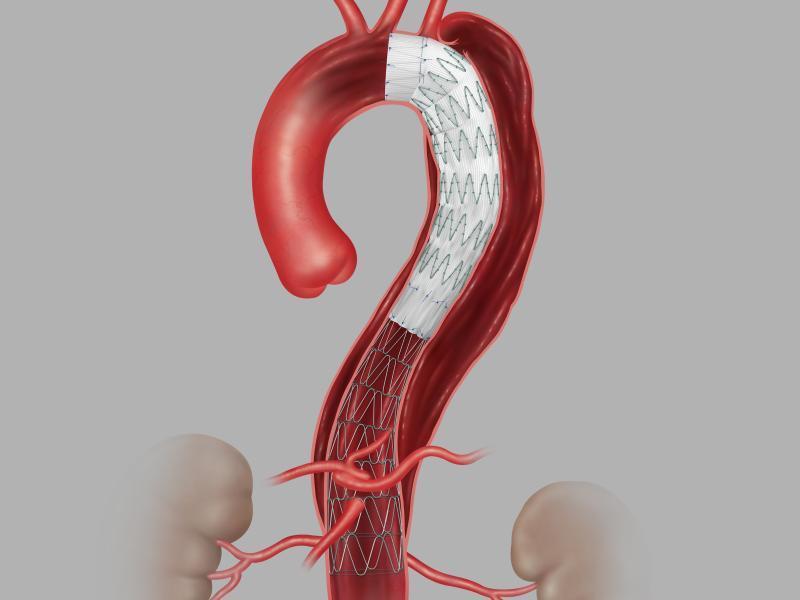
The Zenith system provides physicians a less invasive alternative to open surgery for repair of Type B dissections of the descending thoracic aorta.
The Aortic dissection is a medical condition where a tear occurs between the innermost and middle layers of the aorta. Following the tear of the inner layer of aorta, blood flows through the tear, making the inner and middle layers of the aorta to separate (dissect).
Cook Medical’s Zenith system consists of a proximal stent-graft component and a distal bare stent component to provide the physicians with less invasive alternative to open surgery for repair of Type B dissections of the descending thoracic aorta. Type B dissection involves a tear in the descending aorta.
Cook Medical vascular division vice president Mark Breedlove said: “We are committed to helping patients by developing a variety of treatment options for aortic disease. We’re pleased to provide US clinicians and patients another minimally invasive option for aortic repair.”
The company said that thoracic endovascular aortic repair (TEVAR) is treatment is usual choice for complicated Type B aortic dissection worldwide. The procedures are meant to prevent malperfusion of aortic branches and aortic rupture.
Cooper aortic center director Joseph Lombardi said: “The value in this dissection stent is that it’s pathology-specific, designed just for this disease. As the principal investigator, receiving FDA approval is something that I had really looked forward to. It’s really exciting and I think it’s going to make a big impact on how dissection is managed.”
Cook Medical has secured the US FDA clearance for the Zenith dissection endovascular system recently in February 2019.
In September 2018, Cook Medical has secured the FDA approval for its 5mm diameter version of Zilver PTX superficial femoral artery (SFA) drug-eluting stent.
The company claims that its Zilver PTX is the first 5mm drug-eluting stent in the US, available in lengths up to 140mm diameter and is indicated to treat vessels as small as 4mm in diameter.
Zilver PTX is produced in various stent diameters for the treatment of vessel sizes ranging between 4mm and 7mm. The 5mm size drug-eluting stent is newly made available in the US market for peripheral artery disease (PAD).






