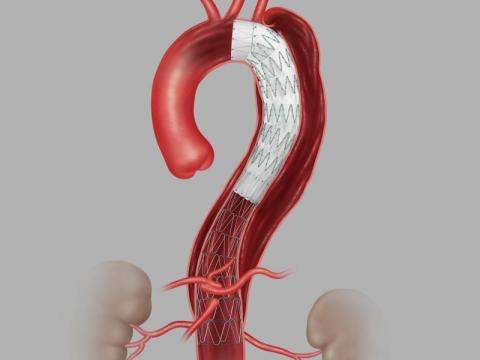
The system, consisting of a proximal stent-graft component and a distal bare stent component, provides physicians a less invasive alternative to open surgery for repair of Type B dissections of the descending thoracic aorta. The device will be available for sale in the U.S. in the coming months.
“We’re pleased to provide another minimally invasive option for aortic repair,” said Mark Breedlove, vice president of Cook Medical’s Vascular division. “The approval of this product gives us an opportunity to have a positive impact on the lives of patients with aortic dissections.”
Aortic dissection is a tear that occurs between the innermost and middle layers of the aorta. When the inner layer of the aorta tears, blood flows through the tear, which causes the inner and middle layers of the aorta to separate (dissect). Type B dissection involves a tear in the lower (descending) aorta, while Type A dissection involves a tear in the upper (ascending) aorta.
Globally, thoracic endovascular aortic repair (TEVAR) is acknowledged as the treatment of choice for complicated Type B aortic dissection. These procedures are meant to prevent malperfusion of aortic branches and aortic rupture.
“Cook Medical is committed to developing a variety of treatment options for aortic disease –from the arch to the iliacs, in order to help physicians fit a device to each patient’s unique disease state,” Breedlove said.
Source: Company Press Release






