Medical technology company Brainlab has secured approval from the US Food and Drug Administration (FDA) for its Loop-X mobile imaging robot and Cirq robotic surgical system for spine.
The approval allows to provide Cirq robotic alignment module for spine procedures in the US. It also enables the introduction of Loop-X, a fully robotic intraoperative imaging device on the market.
The company already secured CE mark approval for both the devices in last summer.
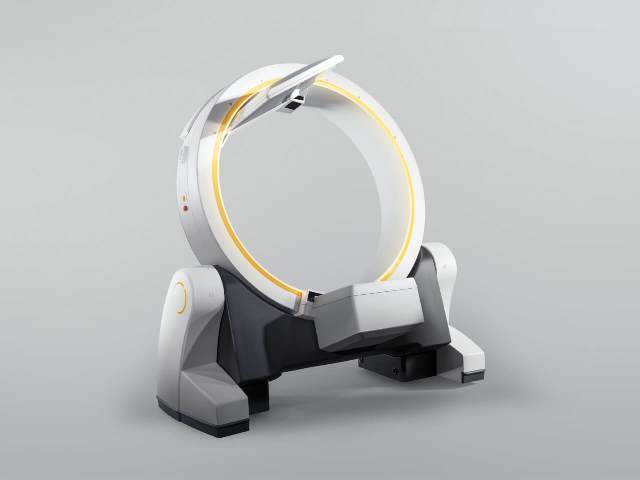
Loop-X, which is claimed to be the first of its kind, is designed to work with the full Brainlab digital surgery portfolio or with a customer’s existing surgical setup.
The mobile imaging robot features an independently moving imaging source and detector panel for flexible patient positioning and non-isocentric imaging.
According to Brainlab, the imaging robot helps in minimising the amount of radiation exposure and increase the variety of indications for the treatment.
Loop-X, which is controlled wirelessly with a touchscreen tablet, has been developed by Brainlab and Austria-based medPhoton.
The world’s first navigated spine surgery using Loop-X mobile imaging robot technology was recently conducted by Spain-based Hospital San Juan de Dios León.
The new Cirq robotic alignment module has been designed to adjust the alignment to a pre-planned trajectory and freeing up surgeons’ hands, thereby helping them to better focus on the patient’s anatomy.
UK-based Royal London Hospital’s surgeons have already used Cirq robotic alignment for a range of cases from routine lumbar fusions to complex deformity and cervical fractures.
Brainlab president Sean Clark said: “We’re expanding and diversifying our digital surgery portfolio with robotics across all indications.
“Our customers want to offer their patients advanced technologies close to home. Brainlab technologies are designed to enable greater freedom for clinicians and enhance outcomes for patients.”
In January 2020, Brainlab acquired US-based VisionTree Software, which is engaged in the development of cloud-based and patient-centric data collection and health management solutions.






