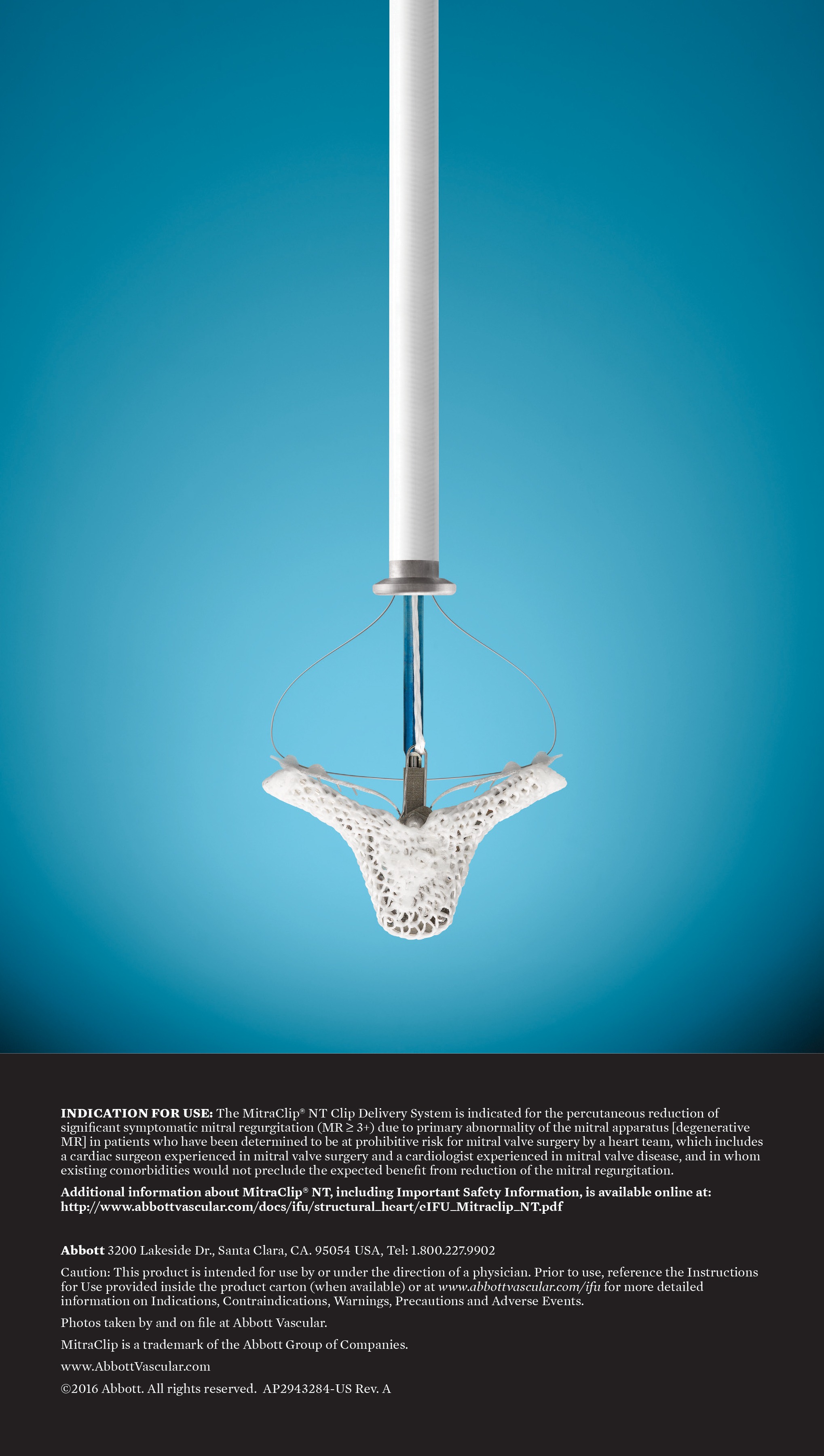
Abbott has received the US Food and Drug Administration (FDA) approval for a clinical study of its minimally invasive MitraClip device in open heart mitral valve surgical repair.
The medical devices firm said that the clinical trial, dubbed REPAIR MR, is designed to evaluate the effectiveness of MitraClip in performing open heart mitral valve surgical repair procedure in eligible people with primary mitral regurgitation (MR).
Abbott structural heart business chief medical officer Neil Moat said: “Abbott is leading the way in the structural heart space. We’re pushing the field forward by making clinical investments to examine whether new, minimally invasive treatment options are suitable, or even preferable, to what has been the standard of care.
“Devices that can be delivered through a minimally invasive method to close or repair a significant structural issue in the heart are in high demand, and we are committed to continuing our efforts to bring the benefits of these devices to patients who need them.”
MitraClip system demonstrated positive results for both primary and secondary MR
Mitral regurgitation (MR) is a progressive debilitating disease, characterised by the incomplete closing of the heart’s mitral valve, leading to backward flow and leak of blood into the atrium of the heart. MR contributes for reduced quality of life, recurrent hospitalisations and decreased survival.
Abbott said that its MitraClip system has shown positive impact for patients with both primary and secondary MR. Its advanced MitraClip therapy is said to improve clinical outcomes and quality of life through a minimally invasive option that reduces severe symptoms of MR.
The MitraClip system has been commercially available in the US since 2013 and in Europe since 2008.
The REPAIR MR clinical trial is planned to enrol approximately 500 patients from 60 sites across the US, Canada and Europe to assess the effectiveness of the MitraClip device in patients with severe primary MR, who are eligible for open-heart surgery.
REPAIR MR trial co-principal investigator Patrick McCarthy said: “The REPAIR MR trial seeks to evaluate the MitraClip device in treating a new patient population who currently undergo the standard surgical treatment, but are at moderate surgical risk.
“This is an important question since approximately 70% of people diagnosed with primary mitral regurgitation aren’t treated with open-heart mitral valve surgery today yet are in need of treatment and symptom relief.”






