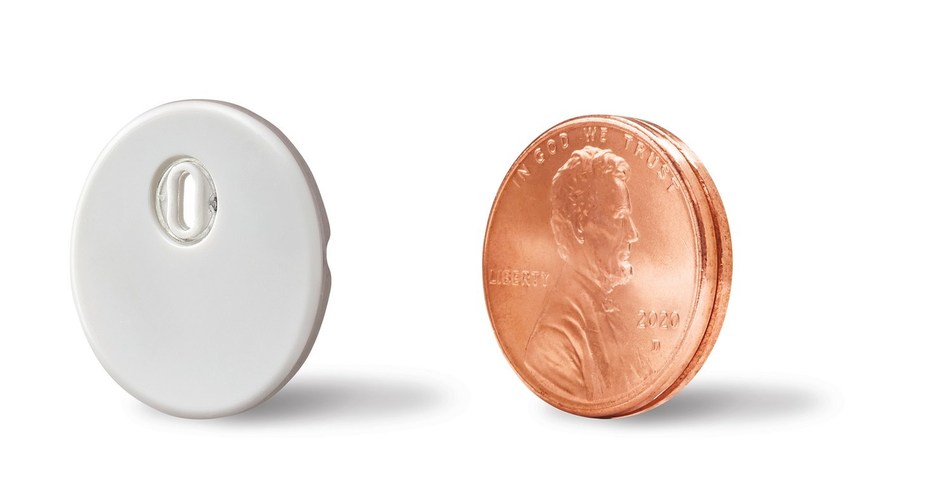
Abbott has received CE Mark approval for its new-generation FreeStyle Libre 3 system, to offer continuous glucose monitoring (CGM) for people with diabetes in Europe.
The new device is designed to continuously measure the glucose readings in real-time, and automatically deliver the readings to smartphones, every minute.
The FreeStyle Libre 3 System features a small and thin sensor that can be easily worn on the back of the upper arm using a one-piece applicator, and can be continuously worn for up to 14 days.
The advanced sensor prevents the need for painful finger sticks to measure glucose levels.
The medical devices company said that the self-applied CGM sensor is the longest-lasting sensor available, and is offering the system at the same price as its previous versions, to allow more people with diabetes access the advanced technology.
Abbott diabetes care senior vice president Jared Watkin said: “Abbott won’t stop innovating when there’s room to raise the bar. We’ve done that again with FreeStyle Libre 3, the smallest sensor that delivers life-changing benefits and best-in-class accuracy.
“People living with diabetes are at the center of our design process, and we made our next-generation technology even more discreet for a better user experience to make managing diabetes as easy and seamless as possible.”
Apart from the sensor, the CGM system includes the FreeStyle Libre 3 mobile app, designed to capture and view real-time glucose levels, glucose history and trend arrows, enabling users know their changing glucose levels using smartphone.
FreeStyle Libre 3 system’s compact sensor design contributes to the sustainability
Abbott said that the use of smaller sensor in next-generation FreeStyle Libre 3 system is aimed at reducing the total volume of the system by more than 70%, to be environment friendly.
The new sensor design reduces use of plastics by 41% and carton paper by 43%, and is said to complement the company’s sustainability targets.
Abbott intends to commercialise the FreeStyle Libre 3 system in Europe in few months.
Furthermore, the company’s FreeStyle Libre products are said to improve glucose control, increase time target glucose range, reduce hyperglycemia and hypoglycemia, and HbA1c, contributing to better health outcomes.
Watkin added: “Since we launched the first disposable glucose sensor in 2014, we’ve always believed all people with diabetes should have access to high-quality, accurate and affordable diabetes technologies.
“That’s why Abbott disrupted the traditional notion that CGMs have to sacrifice quality or accuracy for affordability, and we built our FreeStyle Libre family of products to deliver unparalleled results at a lower cost than any other CGM available.”






