As the OEM division of Merit Medical Systems, our function is to partner with designers, entrepreneurs, individuals, and companies who are designing a new device or changing the functionality of a device. Many OEM customers prefer to use Merit OEM as a Private Labeler or Virtual Manufacturer, trusting in our processes of design, validation, production, manufacturing, packaging, and worldwide transportation. Customers begin with the Merit OEM catalog containing thousands of field-proven medical devices and components, and work with our dedicated OEM Engineers and Business Managers to customize these devices or components to meet specific project needs. Finished products can include custom logo printing on devices, custom tray setup, custom packaging, and custom labeling. Discover more at: https://meritoem.com/
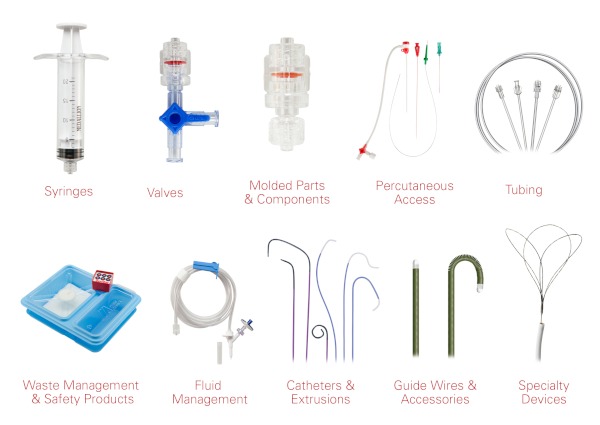
Capabilities Overview
INJECTION MOLDING
• Injection Molding
• Over 60 Injection Molding Machines
• 100% American Made Molds
• Insert Molding
• Two-Shot Molding
COATINGS
• PTFE Coatings
• Reinforced PTFE Coatings
• Hydrophilic Coatings
• Wire Coiling
• Continuous Spool-to-Spool Coatings
• Coated Guidewires and Components
• Coated Mandrels, Stylets & Needles
PRESSURE SENSORS
• Pressure Sensors specialized for medical
monitoring needs
• Electronic Device Assembly
• Ceramic Wafer Manufacturing
EXTRUSIONS
• Custom Catheter Lengths & Shapes
• Catheter Assembly
• Wire Braid Extrusions
• RF Tipping
• Excimer Laser Drilling
• Crimping & Swaging of Radiopaque Marker Band
CLEANROOM ASSEMBLY
• Class 100,000 Cleanrooms
• Over 400,000 ft2 of Cleanroom Space
• Inline Quality Control
• Product Testing
• Automated & Manual Assembly
• UV & Solvent Bonding
• Regulatory Support
• Ultra Sonic Welding
• RF Welding
• RF Tipping
• Guide Wire Assembly
• Bevel Grinding
• Needle Assembly


