The approvals allow Subtle Medical to commercialize the technology across the US and European Economic Area.
The SubtlePET AI solution has been developed to optimize images from faster scans in the hospitals and imaging centers. It will help improve patient experience during imaging procedures and enhance exam throughput and provider profitability.
At present, SubtlePET is in pilot clinical use in various university hospitals and imaging centers in the US and overseas.
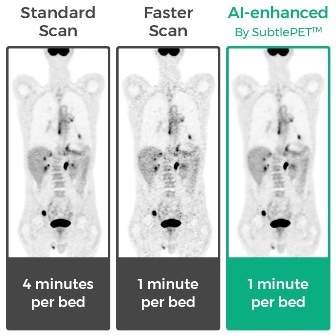
According to the company, the new AI solution allows to complete more exams in a day compared against conventional PET imaging, enabling to reduce capital expenditures.
The SubtlePET is also said to reduce patient time in the scanner, helping hospitals and imaging centers to complete the task rapidly.
SubtlePET takes the support of deep learning algorithms to incorporate with any OEM scanner and PACS system to enhance images during acquisition without any without any disruption or alteration in the imaging workflow of specialists.
The new AI solution will also help in enhancing the image quality of noisy images capturing from shorter scans, which is an advantage for children and those undergoing repeat PET exams.
Subtle Medical founder and CEO Dr Enhao Gong said: “The accomplishment of having the first AI cleared for use in nuclear medicine applications validates our team’s strength and the commitment of our collaborators.
“Our focus on image acquisition and workflow differentiates us from other AI companies that are working on post-processing and computer-aided diagnosis products.
“We are not replacing radiologists–we are addressing the tremendous cost to U.S. healthcare by leveraging deep learning in imaging at the infrastructure level to enable better and higher quality care.”
Subtle Medical is also engaged in the development of additional products, which will be submitted for FDA approval.
The firm’s SubtleMR is currently under clinical assessment, which is being developed to help imaging centers to enhance MRI scans. In addition, SubtleGAD is under development for reduction of gadolinium dosage during imaging procedures.






