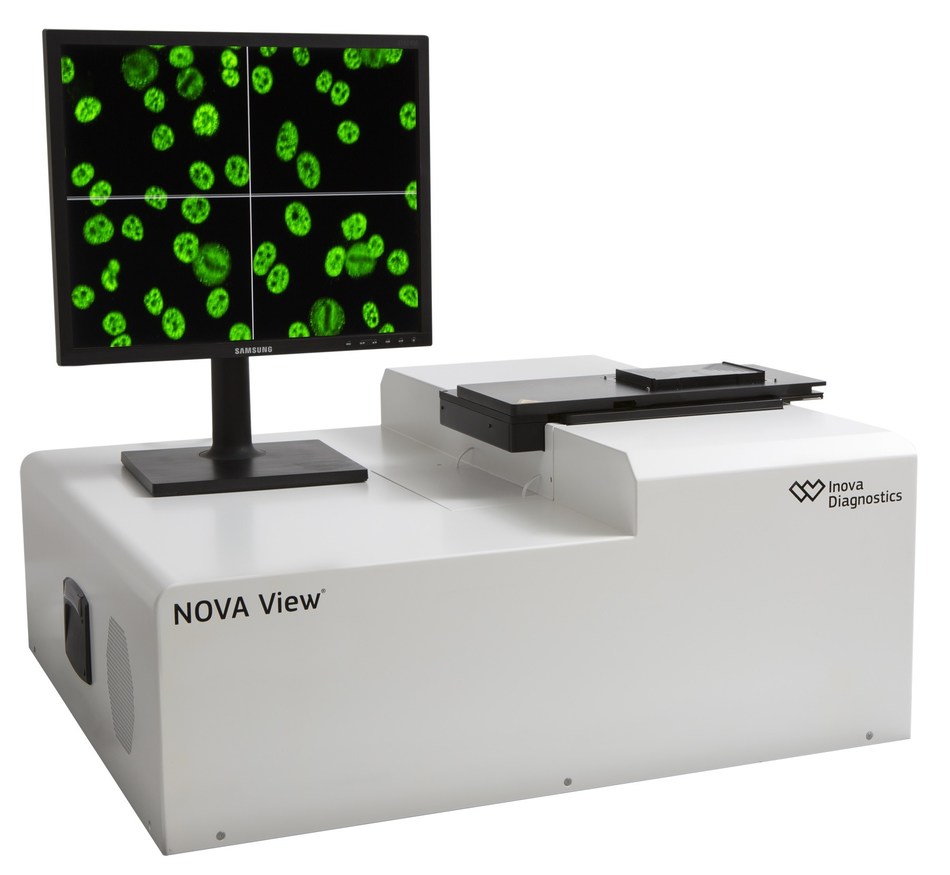
Inova Diagnostics, a world leader in autoimmune disease diagnostic systems and reagents for the clinical laboratory, is pleased to announce the 510(k) clearance of the NOVA Lite® DAPI dsDNA Crithidia luciliae (CLIFT) Kit by the US Food and Drug Administration (FDA). This kit aids in the qualitative and/or semi- quantitative determination of anti-double stranded DNA (dsDNA) IgG antibodies with use of the NOVA View Automated Fluorescence Microscope or manual fluorescence microscopy. The presence of anti-dsDNA antibodies can be used in conjunction with other serological and clinical findings to aid in the diagnosis of systemic lupus erythematosus (SLE)
In SLE, the immune system attacks its own tissues, affecting multiple organs. The classic triad of fever, joint pain, and rash should prompt investigation into the diagnosis of SLE. Diagnosis of SLE ensures proper preventative and medical care to improve quality of life.
Automation using NOVA View and the NOVA Lite DAPI dsDNA Crithidia luciliae Kit can significantly improve laboratory turnaround time and reduce technician hands on time with the detection and titering of anti-dsDNA antibodies. The single well titer application contributes to these workflow and time savings by reducing the number of wells required to perform anti-dsDNA antibody testing.
“Inova Diagnostics is thrilled to add an automated solution for detection of anti-dsDNA IgG antibodies that aid in the diagnosis of SLE. CLIFT is a well-established and highly specific assay capable of detecting high avidity IgG antibodies,” says Kishore Malyavantham, Director of Research and Development at Inova Diagnostics. “This kit expands the offerings on the NOVA View Automated Microscope and is a step forward in helping laboratories streamline their workflow and achieve accurate results.”
Source: Company Press Release






