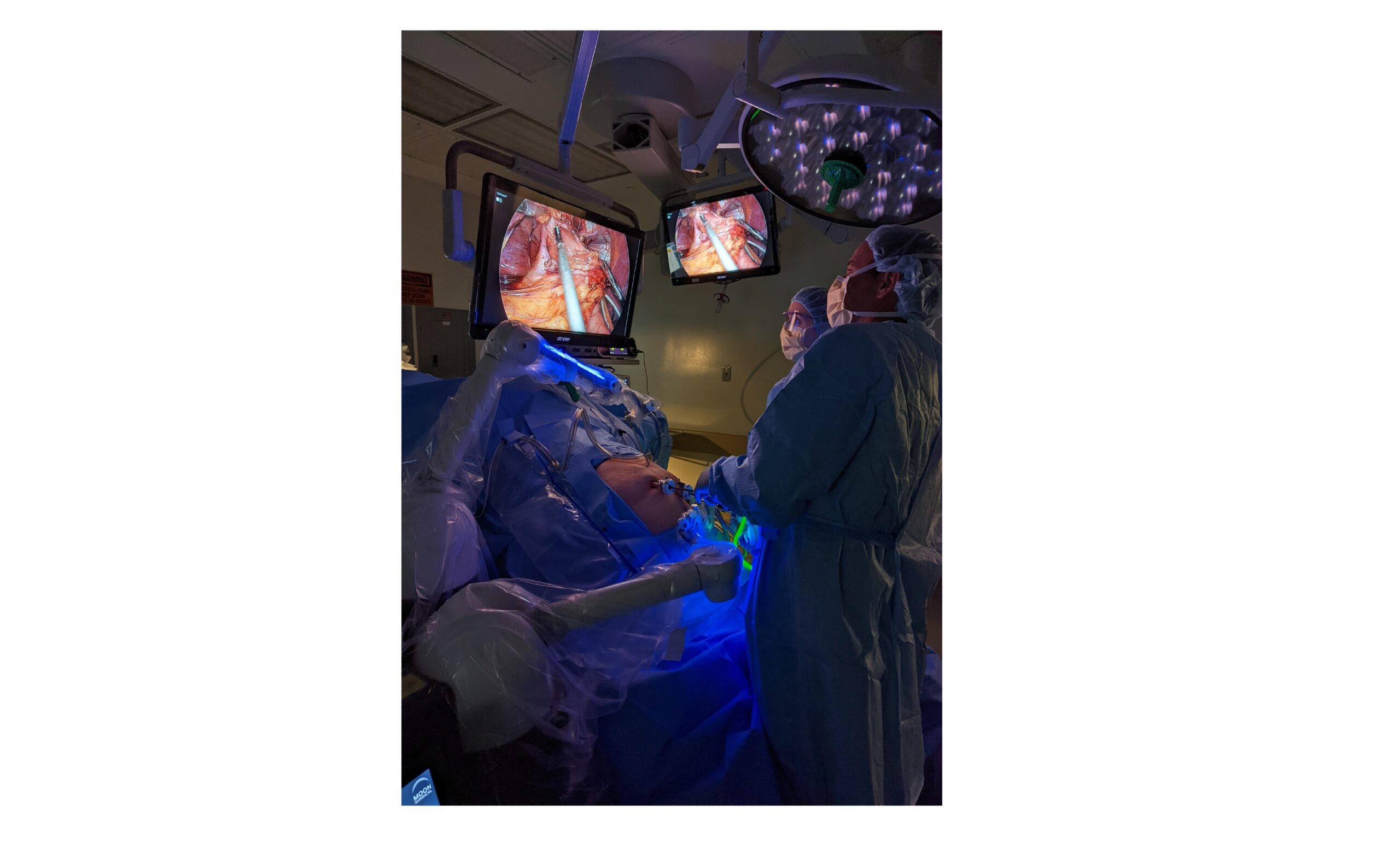
Moon Surgical, a French-American pioneer in collaborative robotics, today announced that Baptist Health (Jacksonville, Florida) performed the first clinical procedures in the United States using the Maestro System.
Three surgeons who practice at Baptist Health hospitals used the Maestro System over a week to perform sleeve gastrectomies – the most common bariatric (weight loss) surgery. The surgeons employed by North Florida Surgeons included Steven Hodgett, MD, FACS; Craig Morgenthal, MD, FACS; and Alexander Rose, MD.
“The Maestro System is an asset to my laparoscopy practice,” said Dr. Hodgett, Medical Director, Baptist Center for Bariatric and Reflux Surgery. “During procedures, the two co-manipulative arms provided me with stable vision and dependable organ retraction, allowing me to give more focus to my surgical case and perform at my best.”
An important added benefit is the ability of the system to reduce the number of operating room staff required to support a laparoscopic procedure. Maestro takes on the tasks of holding and adjusting instruments as needed, empowering hospitals to optimize their OR staff distribution, and surgeons with greater control, confidence, and efficiency.
“This technology is coming to us at a great time,” states Ron Landmann, MD, Chief of Colon & Rectal Surgery and Medical Director of Informatics at Baptist Health. “Maestro provides the control and dependability needed in minimally invasive surgery at a time when staffing shortages introduce workflow challenges and variability.”
The Maestro System was designed with ease of use at the forefront; allowing for minimal training and rapid deployment in surgical departments.
“After treating fifty patients in our First-in-Human clinical trial, we are excited to be doing clinical cases in the US with a world-class institution like Baptist Health,” said Anne Osdoit, CEO of Moon Surgical and a Partner at Sofinnova Partners. “Their commitment to advanced technology that delivers real value to their patients and their staff is well recognized.”
The company received its first 510(k) clearance in December 2022.
Source: Company Press Release






