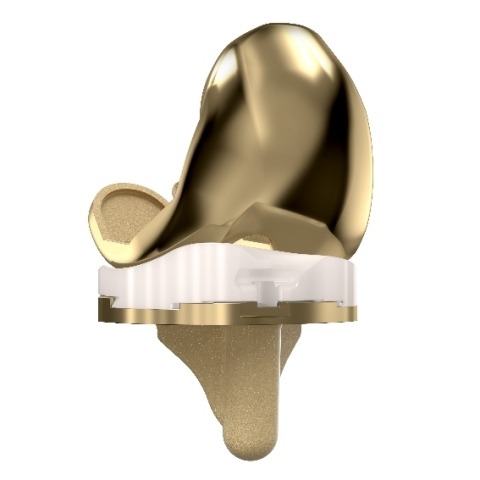
The MicroPort Orthopedics medial-pivot knee system now stands on over 20 years of clinically demonstrated success with its novel medial-pivot design built on the evidence of the natural stability and kinematic motion of the knee.
The Evolution NitrX Medial-Pivot Knee features a titanium niobium nitride (TiNbN) coating that has been shown in simulated clinical testing to reduce the release of cobalt (Co), chromium (Cr), molybdenum (Mb), and nickel (Ni) ions common in standard CoCr implants.
The Evolution NitrX Medial-Pivot Knee builds upon the same medial-pivot legacy of 95% patient satisfaction with 98.8% survivorship at 17 years.
“I see a dramatic difference in patients treated with the Evolution Medial-Pivot Knee in terms of pain and recovery. When I see these patients at four or six weeks, they’re just at a different stage of recovery than I was seeing with my previous patients treated with traditional implants,” says Russell Nevins, MD, Fellowship-Trained Orthopedic Surgeon at Desert Orthopedic Center in Las Vegas, NV.
“With the Evolution NitrX knee, we now have the ability to deliver the benefit of the medial-pivot design to a broader array of patients.”
Established in January 2014, MicroPort Orthopedics is the fourth largest multinational producer of orthopedic products and a proud member of the MicroPort Scientific Corporation family of companies.
From its headquarters in Arlington, Tennessee, MicroPort Orthopedics develops, produces, and distributes innovative OrthoRecon products. The company’s U.S.-based manufacturing and logistics capabilities deliver high quality hip and knee products to patients and their doctors in over 60 countries, including the U.S., EMEA, Japanese, Latin American and China markets.
Source: Company Press Release






