Participating in the current round is the leading dialyzer manufacturer in Japan, Asahi Kasei Medical, as well as the American investment company Tal Capital, and private investors such as Mickey Boodaei, Shai Agassi, Yuval Tal and Meir Barel.
They join the company’s existing investors, who include Chinese pharma giant Haisco, Nava and Yehuda Zissapel, Zohar Gilon, Eri Steimatzky, Henit Vitos, and Ari Raved. Laminate was founded in 2012 by Tammy Gilon, who is the company’s CEO, and Dr. Eyal Orion.
A few months ago Laminate began the stage of clinical trials in the USA, and is working with 16 leading American hospitals in preparation for obtaining FDA approval. The product developed by the company, the VasQ, already has CE approval in Europe.
Through Laminate’s local operation, it is in use in dozens of hospitals in Germany (and is included in the insurance companies’ approved reimbursements). Laminate has significantly expanded its activities in the German market following the approval of additional reimbursement coverage in 260 hospitals in the country.
It is also sold through a network of distributors in other countries, such as Italy, Sweden, and Austria. In Israel the device is in use in two hospitals, Sheba and Hadassah Ein Kerem, with impressive results.
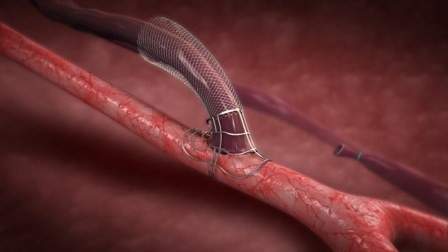
Laminate’s VasQ device is intended for patients suffering from end stage renal disease and requiring dialysis. Prior to this a fistula operation is required, in which a blood vessel connection is made between a vein and an artery, usually in the area of the forearm or elbow. Two intravenous needles are inserted through this connection, to remove the patient’s blood for filtering in the dialysis machine, and then return it.
This is necessary to ensure a sufficiently high circulation of blood, and enable a large volume of blood to be transferred, since the vein is unable to carry the required volume, while the arteries are too deep for repeated insertion of a needle. However, given the high rate of failure of these operations, a solution is required to enable renal failure patients to undergo dialysis without repeated interventions every few months, affecting the treatment.
Today, a venous blockage occurs in over half the cases as a result of differences in venous wall tension, and the body’s response of thickening the vein, affecting blood flow during dialysis. This means that patients have to have repeat surgery, affecting the treatment and creating pressure on the hospital resources.
The special device developed by Laminate is a kind of sleeve placed over the vein, which creates the optimal geometrical configuration with the artery, and reduces the tension in the vein, thus enabling appropriate blood flow during dialysis. Research carried out to date indicates significant success in all accepted parameters in the performance of the fistulas.
“After the success of treatments in Europe, including application of the device in hundreds of patients in Germany and other European countries, we are now preparing to enter the American market. We have completed the infrastructure for this, and are working in cooperation with leading hospitals across the USA to carry out the necessary clinical trials for FDA approval,” says Tammy Gilon, Co-Founder and CEO of Laminate.
“The current round is an important expression of faith on the part of the existing investors, and a significant boost from investors with experience in a variety of fields, who understand the need for the solution and the answer that it provides for dialysis patients. Furthermore, the fact that Asahi Kasei Medical has joined us as an investor in the company gives us an important advantage for a future entry into the Japanese market.”
Takeshi Himeno, the president of Asahi Kasei Medical, says “We are so happy to be a part of the company’s journey. I believe VasQ has a high potential to become the standard of care for vascular access surgery.”
Source: Company Press Release






