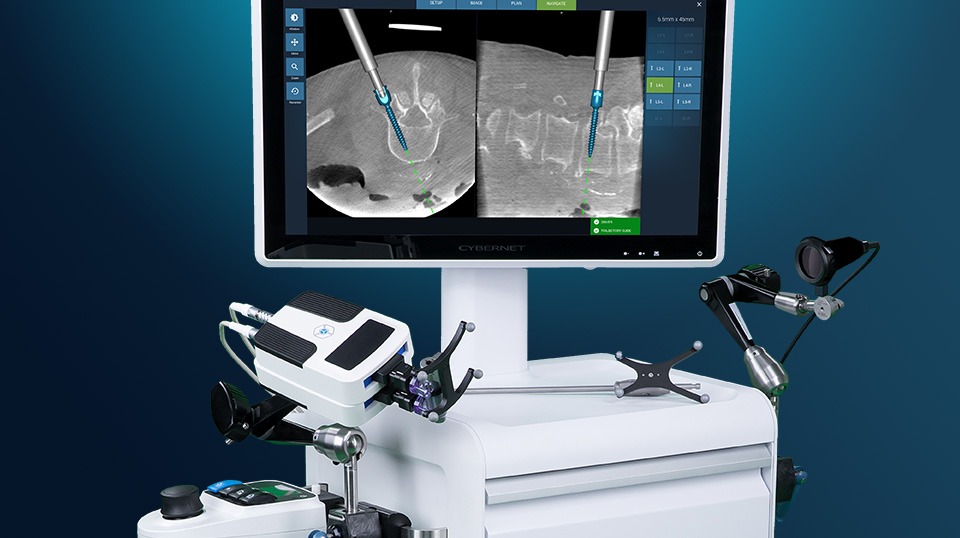
Spinal robotics firm Fusion Robotics has received FDA 510(k) clearance to market their 3D-imaging compatible navigation and robotic targeting system for spinal surgery in the US.
According to the company, the Fusion Robotics System addresses the key limitations of current spinal navigation and robotics systems by offering greater procedural efficiency with significantly less expense.
Fusion Robotics CEO Brad Clayton said: “We appreciate the partnership developed with FDA to rigorously validate safety and accuracy.
“Next, we look forward to partnering with clinicians and hospitals to increase efficiency, reduce cost and broadly expand the application of robotics to treat patients.”
Fusion Robotics chief medical officer Kevin Foley added: “Since the introduction of robotics in spine surgery, the questions of economics and efficiency have hindered the widespread adoption of this technology.
“With this clearance, and the rapid development of our fluoroscopy-integrated system, we’re working quickly towards making robotics available to many more surgeons, in a form better suited to their practices.”
The company plans to commercialise broadly across the US in 2021.
How does Fusion Robotics System improve on existing technology?
The Fusion Robotics System is a small, robotic device that works in tandem with a navigation system to assist surgeons in placing pedicle screws – small screws placed above and below vertebrae that strengthen spinal fusion sites.
According to an interview given by Clayton to CIO Review, setting up the robot takes ten minutes as opposed to the 30 to 40 minutes needed to prep existing robotics technology used for the same purpose.
These existing products, according to him, cost four times the amount of the Fusion Robotics System and weigh close to 400lbs compared to his company’s product, which weighs 4lbs.
FDA 510(k) approval is given to products that are similar to others already on the market as long as a company can demonstrate that it is it just as safe and effective, and works in the same way.






