The prospective, multi-center, two-arm trial plans to enroll 668 patients undergoing treatment for a STEMI heart attack. Half the patients will be randomized to receive delayed reperfusion after 30 minutes of left ventricular unloading with the Impella CP. The other half will receive immediate reperfusion, the current standard of care.
The trial will test the hypothesis that unloading the left ventricle for 30 minutes prior to reperfusion will reduce myocardial damage from a heart attack and lead to a reduction in future heart failure related events. Myocardial damage can lead to an infarct, and every 5% increase in infarct size is associated with a 20% increase in relative hazard for all-cause mortality or hospitalization for heart failure within one year after a primary PCI1. Coronary artery disease is the number one cause of death in the United States. 47% of women and 36% of men over the age of 45 will die within 5 years of their first heart attack.
“The STEMI DTU Pivotal Trial has the potential to improve the standard of care, slow the growing epidemic of heart failure and improve outcomes for millions of heart attack patients. This trial is the first of its kind to focus on ventricular unloading as part of a therapeutic approach for heart attacks without cardiogenic shock and could lead to a paradigm shift in the way heart attack patients are treated worldwide,” said Navin Kapur, MD, the study’s co-principal investigator and the executive director of the CardioVascular Center for Research and Innovation (CVCRI) at Tufts Medical Center.
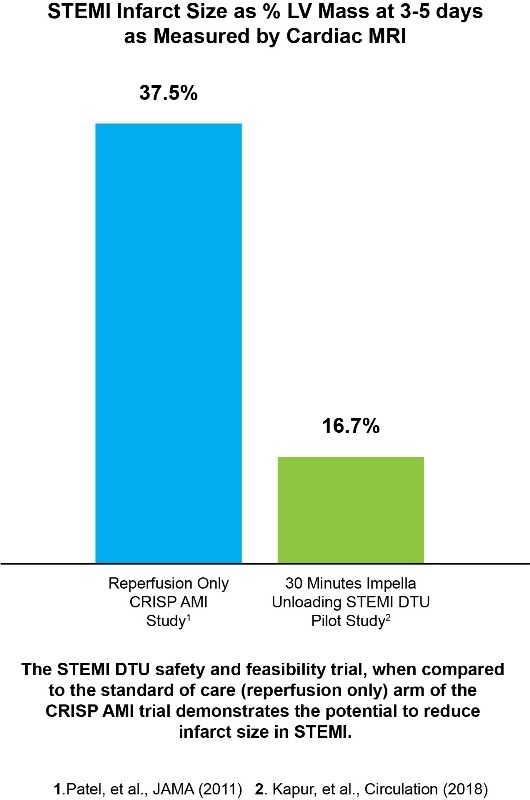
The pivotal trial will build on the promising results of the successful STEMI DTU safety and feasibility trial, which met its goal by demonstrating it is safe and feasible to conduct a study of 30 minutes of unloading with delayed reperfusion. The accompanying chart compares the safety and feasibility trial results to the CRISP AMI trial’s standard of care arm (reperfusion only) and demonstrates how 30 minutes of unloading prior to reperfusion has the potential to reduce infarct size in STEMI.
“A successful study will transform the treatment of heart attacks and reduce heart failure for hundreds of thousands of patients each year. As a result, it is important to execute with the highest scientific rigor to ensure a successful study concludes with a Class I recommendation to reach 200,000 U.S. heart attack patients and more than 4 million patients outside the U.S.,” said Michael R. Minogue, Chairman, President and CEO of Abiomed.
The pivotal trial will be overseen by a steering committee of five expert cardiologists and clinical trialists. They are: Dr. Kapur, William O’Neill, MD, medical director of the Center for Structural Heart Disease at Henry Ford Hospital, Jeffrey Moses, MD, professor of medicine at Columbia University Medical Center and director of interventional cardiovascular therapeutics at Columbia University Medical Center, Gregg Stone, MD, professor of medicine at Columbia University College of Physicians and Surgeons and director of cardiovascular research and education at the Center for Interventional Vascular Therapy at New York-Presbyterian Hospital/Columbia University Medical Center, and James Udelson, MD, chief of the division of cardiology at Tufts Medical Center.
The trial is sponsored by Abiomed. Impella heart pumps are not FDA approved for use in STEMI patients without cardiogenic shock.
Source: Company Press Release.






