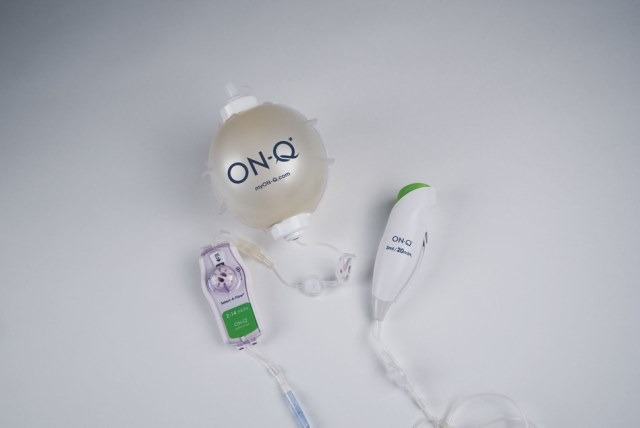
The new ON-Q with Bolus design come with improvements that make easier for providers and patients to use, and also helps to reduce postoperative opioid use to achieve pain management.
The new ON-Q Bolus, which is similar to the original ON-Q pump bolus device, offers customized control for patients to recover from post-surgical pain by delivering continuous and non-opioid medication to the surgical site or peripheral nerves for up to five days.
With an easy to remove priming tab and large level indicator markings, the bolus pump is ergonomically designed to fit comfortably in the patient’s hand.
The new bolus pump design can also be used for incisional applications, offering an added advantage to the surgeons.
According to the company, studies have demonstrated that using bolus rates as low as 3ml in addition to the basal rate can reduce the total amount of anesthetic required for pain relief.
The firm has developed ON-Q to considerably reduce opioid use for surgical patients and provide better pain relief than opioids alone, enabling patients to become normal after surgery.
Avanos Medical said that the new ON-Q Bolus pump will be launched soon into the market.
Avanos CEO Joe Woody said: “The changes we have made to the ON-Q Bolus pump are a direct result of the feedback we heard from our customers.
“Non-opioid approaches to pain management continue to be an important issue in the healthcare industry, particularly in light of the opioid crisis. As a leader in non-opioid treatment for acute pain, we are committed to continuously improving our ON-Q pumps to deliver increased quality of life and satisfaction to all patients.”
Based in Alpharetta of Georgia, Avanos Medical is focused on delivering clinically advanced breakthrough medical device solutions to improve patients’ quality of life.
Avanos is involved in the development, manufacturing and marketing of its brands in more than 90 countries.






