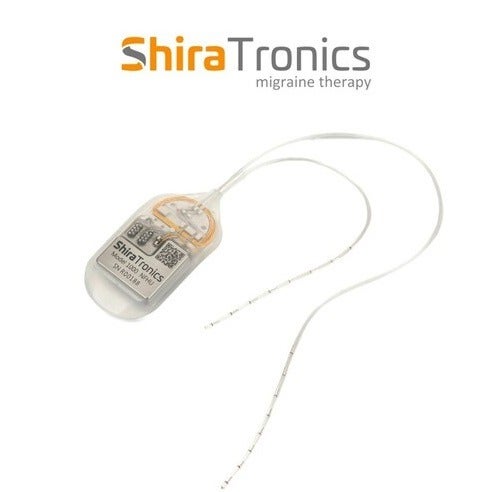
US-based medical device company ShiraTronics has implanted its ShiraTronics Chronic Migraine System in the world’s first six patients.
ShiraTronics said the device is the first fully implantable system for head location that uses a minimally invasive method for neuromodulation.
The system delivers precise electrical pulses, which are customised to interfere with migraine pain signals.
The fully implantable, programmable device provides doctors and patients with a fresh, possibly more efficient therapy option.
ShiraTronics migraine system secured the US Food and Drug Administration (FDA) Breakthrough Device designation in 2021.
The system is intended specifically for migraine symptoms faced by pre-menopausal women.
According to the medical device firm, the device is an alternative to existing migraine treatment that offers limited relief.
ShiraTronics co-founder Lynn Elliott said: “It is gratifying and humbling to witness the evolution of this innovative approach to treating chronic migraine.
“Originating as a visionary concept by a creative group of physicians, it has now materialised into a tangible reality that has the potential to restore quality of life to many enduring this debilitating condition.”
ShiraTronics is collaborating with Australian academics, neurologists, and pain management doctors to undertake the RELIEV-CM clinical study of the ShiraTronics device and gather relevant data.
The study lays the groundwork for further clinical investigations and will assess the effectiveness of the ShiraTronics Chronic Migraine System.
ShiraTronics CEO Rob Binney said: “The achievement of this crucial milestone has validated all aspects of our procedure, system, and therapy settings.
“Notably, it is encouraging that all six patients who completed the trial procedure opted for the permanent implantation of the ShiraTronics system.
“We are thankful for the invaluable guidance and contributions by our investigators to date and look forward to additional patient implants currently scheduled in the new year.”






