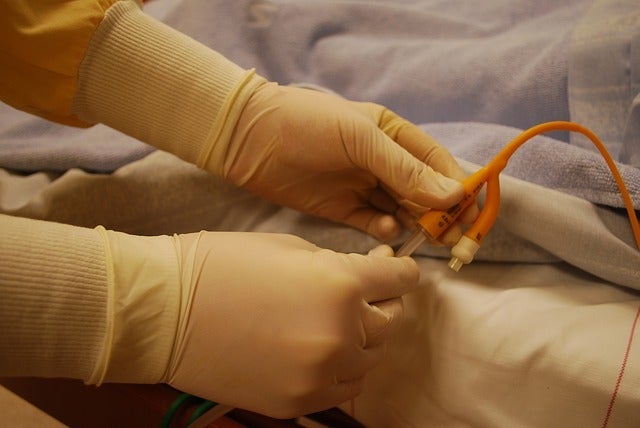
Pathen Medical, a leader in the design and production of specialized medical devices, today announced immediate availability of its NanoCath™ NICU-Comfort™ I.V. Catheter. The new catheter is designed for the smallest patients, including infants in neonatal intensive care units. It features a 26-gauge safety needle containing a higher nickel content for added sharpness and better edge holding, resulting in easier insertion, less pain and improved patient outcomes.
The NanoCath NICU-Comfort catheter material is pressure rated to 300 psi and maintains a fast flow rate of 10 mL/min. The patented catheter material is soft, but has a high tensile strength for easier insertion and a smoother threading feel. Early field adoption has been extremely positive.
“This is an excellent option for the neonate population,” said one RN who participated in field trials. “This device is a long-awaited solution to the difficult problem of neonate vascular access. We have had excellent success with numerous patients and the 26-gauge IV catheter from Pathen rounds out our vascular access toolbox.”
Additional features of the NICU-Comfort Catheter include:
Catheter and hub made from an innovative new material that is softer and more comfortable against the patient’s skin; catheter retains its flexibility to move and bend with the patient without occluding.
Safety IV catheter design to create fast flash and automatic retraction reducing the risk of needle sticks to those inserting the device.
The combined needle and catheter design creates a better threading feel during cannulation and has a longer safe indwell rating.
“This catheter is purpose-built to be a solution for our neonatal providers and their patients,” said Tim Caldwell, founder of Pathen Medical. “Our NICU-Comfort catheter material is soft, reducing kinks and occlusions, and provides greater comfort while remaining highly effective. The flexible material moves with the patient for greater comfort. Combined with our supply-chain innovation, this specialty I.V. catheter is available immediately at a lower price-point than others and without any production or shipping delay. That means better outcomes for the tiniest patients and better results for practitioners.”
As an FDA approved Design Specifier, Pathen Medical has innovated not just in the device but also in the supply chain. Pathen manages a fully operational and uninterrupted supply chain that does not suffer from international manufacturing and export challenges related to material shortages, tariffs or the recent coronavirus outbreak. The company’s focus on quality and innovation also means health care providers are assured a quality product at an affordable price.
Source: Company Press Release






