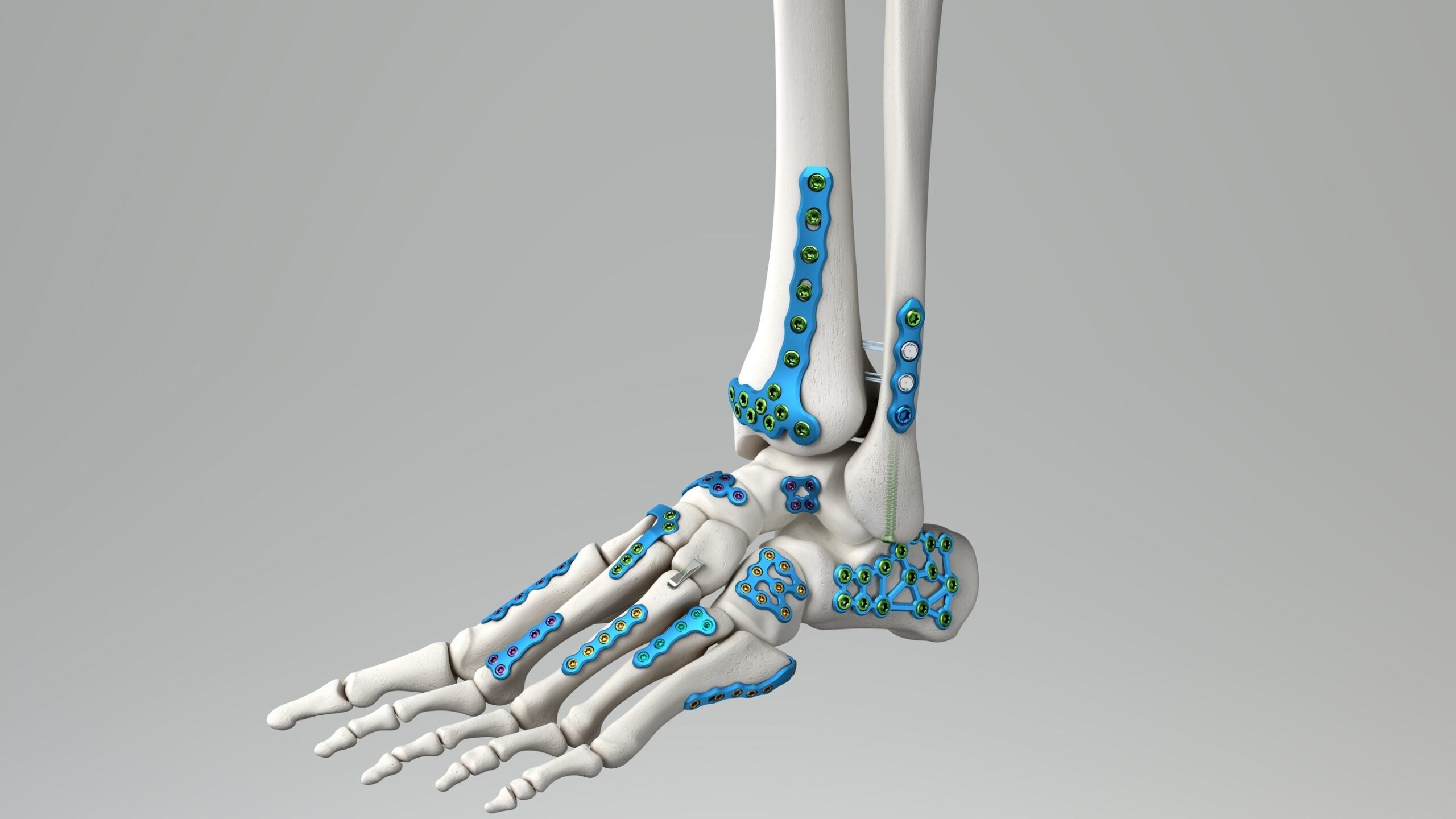
Healthcare firm Medline UNITE Foot & Ankle has introduced the Calcaneal Fracture Plating System and IM Fibula Implant solutions in the US.
The new products will enable surgeons to gain access to a comprehensive titanium foot and ankle trauma system for addressing almost all fractures that require open reduction and internal fixation (ORIF) with plate and screw fixation.
Both solutions have been approved by the US Food and Drug Administration (FDA).
Medline UNITE Foot & Ankle marketing director Scott Goldstein said: “Obtaining FDA clearance and launching these new implant systems comes on the heels of several other recent, successful launches, including the UNITE Jones Fracture Screw System, UNITE Distal Tibia Plating System, and SYNDEX with Constrictor Technology Knotless Syndesmosis Implant System.
“Since Medline UNITE was first established in 2013, we have consistently and intentionally developed systems that offer the latest in implant design and technology, along with unique, bespoke instrumentation, and design rationale continuity across all systems.”
Medline UNITE Calcaneal Fracture Plating System will allow surgeons to handle a wide range of fracture patterns, anatomical variations and surgical methods from one tray.
Many options, including standard, offset, and extension Sinus Tarsi and Perimeter plates are part of the system that also comprises new, fully threaded 5.5mm and 7mm headed cannulated screws.
Furthermore, a Sinus Tarsi Extension Plate Inserter that comes with the system will offer easy insertion and positioning of the plate through the incision.
The IM Fibula Implant provides a new option to surgeons treating transverse fibula fracture patients. It joins the Medline UNITE Distal Tibia System as a standard option.
The patients need intramedullary fixation, which is often linked to distal tibia fractures.
In May last year, Medline agreed to buy a portion of the respiratory business from Teleflex for $286m.






