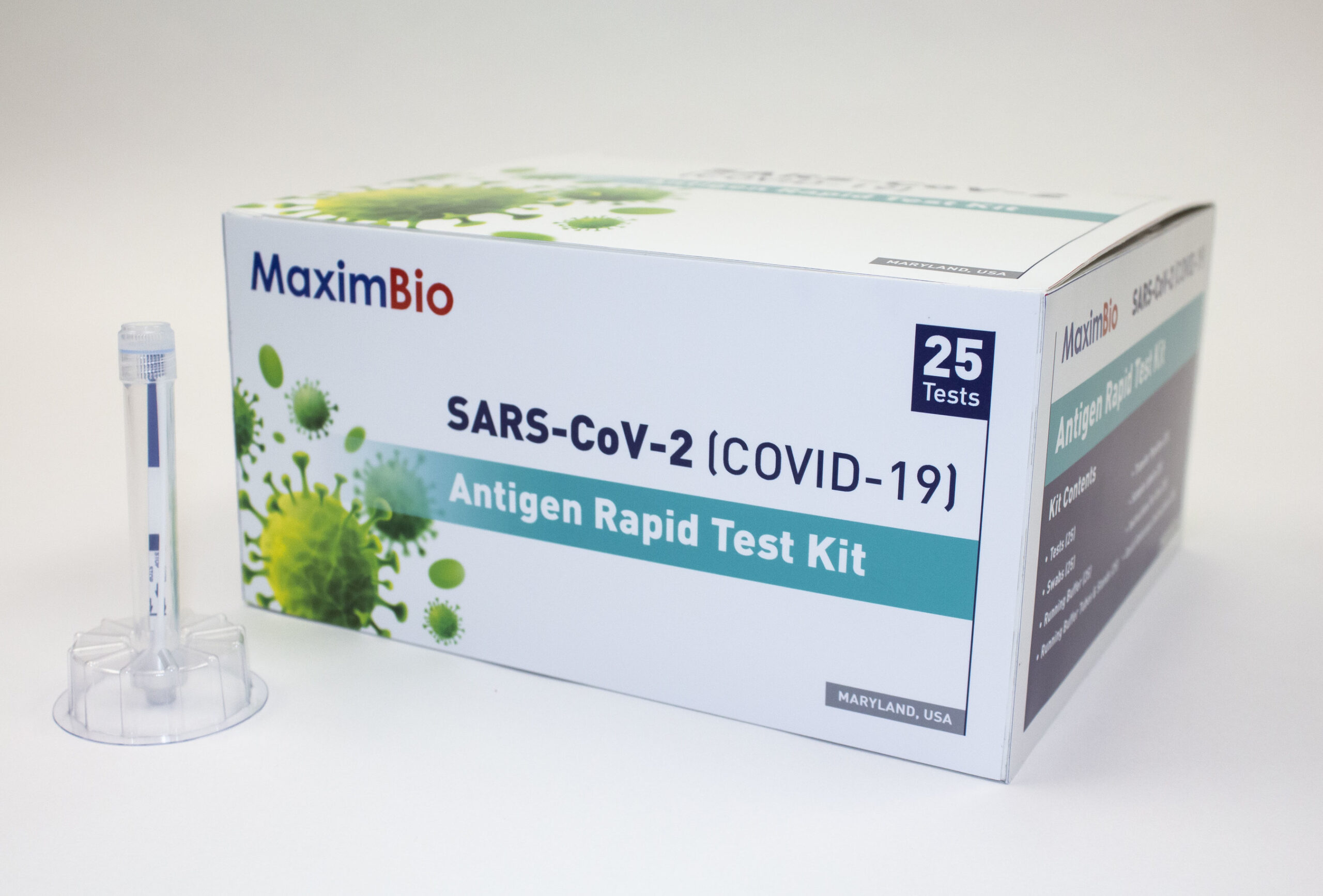
Maxim Biomedical has unveiled its plans to manufacture and commercialise approximately 3 million of its patent pending Maxim SARS-CoV-2 rapid antigen diagnostic tests by the end of 2020.
The company said that it is on track to reach its goal of producing more than one million tests per month by the end of 2020, and achieving a production rate of 15 million tests per month in early 2021.
Maxim Biomedical said that its new rapid antigen test has been developed in collaboration with the National Institutes of Health (NIH), under its rapid acceleration of diagnostics (RADxSM) initiative.
Maxim Biomedical COO Jonathan Maa said: “Maxim Biomedical has always endeavored to provide high quality testing where it would fulfill unmet needs. COVID-19 represents an opportunity to apply our expertise in LFA for application at the point-of-care with an eye to community safety and reducing risk for patients and front-line workers.”
Maxim SARS-CoV-2 rapid antigen diagnostic test provides results within 15 minutes
The Maxim SARS-CoV-2 rapid antigen diagnostic test has been designed to leverage Lateral Flow Assay (LFA) technology with a streamlined workflow, and a closed-tube format to prevent the need for costly equipment or a reader to perform.
Also, the assay is said to combine the specimen swab, reagents, and test strip in a compact self-contained environment, facilitating incubation, reading and disposal in a single tube.
The test provides results within 15 minutes and is stable for more than 2 hours, which makes it a rapid test with result reviewable for a longer time compared to other LFA tests, said the company.
The company is working together with TEAM Technologies to expand manufacturing capacity.
TEAM Technologies president and CEO Marshall White said: “We combined Maxim Biomedical’s development expertise and TEAM Technologies’ specialty manufacturing capabilities to rapidly scale production quantities of the SARS-CoV-2 rapid antigen test. TEAM Technologies is proud to partner with the great folks at Maxim Biomedical to bring this test to market.”
Maxim has partnered with the US Department of Defense, Emory University, Northwestern University and the University of Massachusetts to independently evaluate the assay’s clinical performance.
Emory University School of Medicine paediatrics and biomedical engineering associate professor Wilbur A Lam has led the evaluation of the Maxim SARS-CoV-2 Rapid Antigen Diagnostic Test at Emory.
Lam said: “Our RADx center evaluated Maxim’s technology and our scientists and physicians collectively agreed that this device would be extremely effective. We’re especially enthusiastic about the device’s simplicity, usability, low cost, and convenience.”
Maxim offers a range of FDA and CDC approved products, along with raw materials, and customised CRO/CMO services, through its 26,000ft2 manufacturing facility, which comprises more than 13 individual laboratories.






