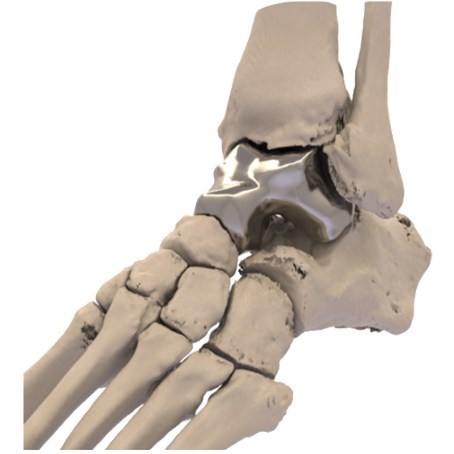
Additive Orthopaedics has secured approval from the US Food and Drug Administration (FDA) for its humanitarian device exemption (HDE) application for the Patient Specific Talus Spacer to treat avascular necrosis of the talus.
The Patient Specific Talus Spacer, which is indicated for Avascular Necrosis (AVN) of the ankle joint, is claimed to be first and only patient specific total talus replacement implant approved by the FDA for use in the US.
Additive Orthopaedics is offering the Patient Specific Talus Spacer as a humanitarian use device.
As defined by the FDA, the device is expected to benefit patients in the treatment or diagnosis of a disease or condition that impacts or is manifested in not more than 8,000 individuals in the US per annum.
The 3D printed and patient specific implant will be designed and produced individually for each patient using CT image data.
According to the company, the device enables a patient to regain motion and reduce pain until the time a fusion potentially becomes necessary.
The company plans to immediately commercialise the Patient Specific Talus Spacer in the US.
Additive Orthopaedics president Greg Kowalczyk said: “Avascular necrosis of the talus is extremely painful and debilitating for these patients. Surgical treatment options are below-the-knee amputation or joint fusion, which results in loss of motion of the ankle and can have poor outcomes.
“The Patient Specific Talus Spacer is another example of how 3D printed devices can improve the standard of care.”
Additive Orthopaedics is said to leverage advanced 3D metal printing technology to design, manufacture and commercialise patient specific and off the shelf implants for complex reconstructions, implant revisions, and limb salvage procedures.






